| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bromodomain-containing protein 3 |
|---|
| Ligand | BDBM50539801 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1979347 (CHEMBL4612482) |
|---|
| Kd | <1.000000±n/a nM |
|---|
| Citation |  Li, Y; Zhao, J; Gutgesell, LM; Shen, Z; Ratia, K; Dye, K; Dubrovskyi, O; Zhao, H; Huang, F; Tonetti, DA; Thatcher, GRJ; Xiong, R Novel Pyrrolopyridone Bromodomain and Extra-Terminal Motif (BET) Inhibitors Effective in Endocrine-Resistant ER+ Breast Cancer with Acquired Resistance to Fulvestrant and Palbociclib. J Med Chem63:7186-7210 (2020) [PubMed] Article Li, Y; Zhao, J; Gutgesell, LM; Shen, Z; Ratia, K; Dye, K; Dubrovskyi, O; Zhao, H; Huang, F; Tonetti, DA; Thatcher, GRJ; Xiong, R Novel Pyrrolopyridone Bromodomain and Extra-Terminal Motif (BET) Inhibitors Effective in Endocrine-Resistant ER+ Breast Cancer with Acquired Resistance to Fulvestrant and Palbociclib. J Med Chem63:7186-7210 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bromodomain-containing protein 3 |
|---|
| Name: | Bromodomain-containing protein 3 |
|---|
| Synonyms: | BRD3 | BRD3_HUMAN | Bromodomain and extra-terminal motif (BET) | Bromodomain-containing protein 3 | Bromodomain-containing protein 3 (BRD3) | KIAA0043 | RING3-like protein | RING3L |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 79571.81 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15059 |
|---|
| Residue: | 726 |
|---|
| Sequence: | MSTATTVAPAGIPATPGPVNPPPPEVSNPSKPGRKTNQLQYMQNVVVKTLWKHQFAWPFY
QPVDAIKLNLPDYHKIIKNPMDMGTIKKRLENNYYWSASECMQDFNTMFTNCYIYNKPTD
DIVLMAQALEKIFLQKVAQMPQEEVELLPPAPKGKGRKPAAGAQSAGTQQVAAVSSVSPA
TPFQSVPPTVSQTPVIAATPVPTITANVTSVPVPPAAAPPPPATPIVPVVPPTPPVVKKK
GVKRKADTTTPTTSAITASRSESPPPLSDPKQAKVVARRESGGRPIKPPKKDLEDGEVPQ
HAGKKGKLSEHLRYCDSILREMLSKKHAAYAWPFYKPVDAEALELHDYHDIIKHPMDLST
VKRKMDGREYPDAQGFAADVRLMFSNCYKYNPPDHEVVAMARKLQDVFEMRFAKMPDEPV
EAPALPAPAAPMVSKGAESSRSSEESSSDSGSSDSEEERATRLAELQEQLKAVHEQLAAL
SQAPVNKPKKKKEKKEKEKKKKDKEKEKEKHKVKAEEEKKAKVAPPAKQAQQKKAPAKKA
NSTTTAGRQLKKGGKQASASYDSEEEEEGLPMSYDEKRQLSLDINRLPGEKLGRVVHIIQ
SREPSLRDSNPDEIEIDFETLKPTTLRELERYVKSCLQKKQRKPFSASGKKQAAKSKEEL
AQEKKKELEKRLQDVSGQLSSSKKPARKEKPGSAPSGGPSRLSSSSSSESGSSSSSGSSS
DSSDSE
|
|
|
|---|
| BDBM50539801 |
|---|
| n/a |
|---|
| Name | BDBM50539801 |
|---|
| Synonyms: | CHEMBL4648912 | US11840533, Compound 70 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H28N6O3S |
|---|
| Mol. Mass. | 552.647 |
|---|
| SMILES | CCS(=O)(=O)Nc1cc(cc2n(ccc12)C(C)(c1ccccn1)c1ccccn1)-c1cn(C)c2c1cc[nH]c2=O |
|---|
| Structure | 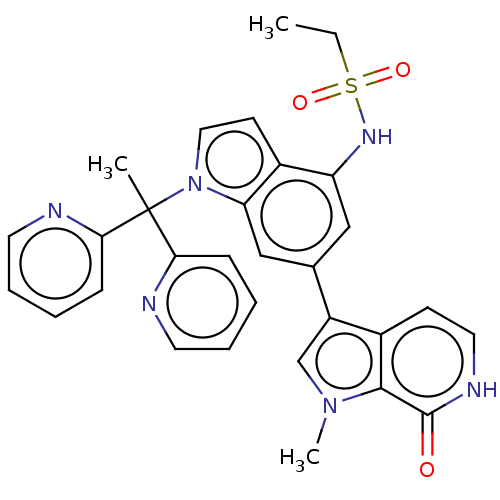
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, Y; Zhao, J; Gutgesell, LM; Shen, Z; Ratia, K; Dye, K; Dubrovskyi, O; Zhao, H; Huang, F; Tonetti, DA; Thatcher, GRJ; Xiong, R Novel Pyrrolopyridone Bromodomain and Extra-Terminal Motif (BET) Inhibitors Effective in Endocrine-Resistant ER+ Breast Cancer with Acquired Resistance to Fulvestrant and Palbociclib. J Med Chem63:7186-7210 (2020) [PubMed] Article
Li, Y; Zhao, J; Gutgesell, LM; Shen, Z; Ratia, K; Dye, K; Dubrovskyi, O; Zhao, H; Huang, F; Tonetti, DA; Thatcher, GRJ; Xiong, R Novel Pyrrolopyridone Bromodomain and Extra-Terminal Motif (BET) Inhibitors Effective in Endocrine-Resistant ER+ Breast Cancer with Acquired Resistance to Fulvestrant and Palbociclib. J Med Chem63:7186-7210 (2020) [PubMed] Article