| Reaction Details |
|---|
 | Report a problem with these data |
| Target | P2X purinoceptor 1 |
|---|
| Ligand | BDBM50540456 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1981892 (CHEMBL4615154) |
|---|
| IC50 | 439±n/a nM |
|---|
| Citation |  Tian, M; Abdelrahman, A; Baqi, Y; Fuentes, E; Azazna, D; Spanier, C; Densborn, S; Hinz, S; Schmid, R; Müller, CE Discovery and Structure Relationships of Salicylanilide Derivatives as Potent, Non-acidic P2X1 Receptor Antagonists. J Med Chem63:6164-6178 (2020) [PubMed] Article Tian, M; Abdelrahman, A; Baqi, Y; Fuentes, E; Azazna, D; Spanier, C; Densborn, S; Hinz, S; Schmid, R; Müller, CE Discovery and Structure Relationships of Salicylanilide Derivatives as Potent, Non-acidic P2X1 Receptor Antagonists. J Med Chem63:6164-6178 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| P2X purinoceptor 1 |
|---|
| Name: | P2X purinoceptor 1 |
|---|
| Synonyms: | ATP receptor | P2RX1 | P2RX1_HUMAN | P2X1 | Purinergic receptor | Purinergic, P2X |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 44992.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Purinergic, P2X 0 HUMAN::P51575 |
|---|
| Residue: | 399 |
|---|
| Sequence: | MARRFQEELAAFLFEYDTPRMVLVRNKKVGVIFRLIQLVVLVYVIGWVFLYEKGYQTSSG
LISSVSVKLKGLAVTQLPGLGPQVWDVADYVFPAQGDNSFVVMTNFIVTPKQTQGYCAEH
PEGGICKEDSGCTPGKAKRKAQGIRTGKCVAFNDTVKTCEIFGWCPVEVDDDIPRPALLR
EAENFTLFIKNSISFPRFKVNRRNLVEEVNAAHMKTCLFHKTLHPLCPVFQLGYVVQESG
QNFSTLAEKGGVVGITIDWHCDLDWHVRHCRPIYEFHGLYEEKNLSPGFNFRFARHFVEN
GTNYRHLFKVFGIRFDILVDGKAGKFDIIPTMTTIGSGIGIFGVATVLCDLLLLHILPKR
HYYKQKKFKYAEDMGPGAAERDLAATSSTLGLQENMRTS
|
|
|
|---|
| BDBM50540456 |
|---|
| n/a |
|---|
| Name | BDBM50540456 |
|---|
| Synonyms: | CHEMBL2313133 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H8ClF6NO3S |
|---|
| Mol. Mass. | 419.727 |
|---|
| SMILES | Oc1ccc(Cl)cc1S(=O)(=O)Nc1cc(cc(c1)C(F)(F)F)C(F)(F)F |
|---|
| Structure | 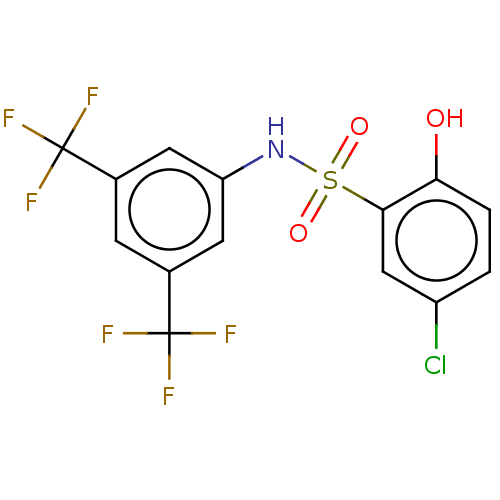
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tian, M; Abdelrahman, A; Baqi, Y; Fuentes, E; Azazna, D; Spanier, C; Densborn, S; Hinz, S; Schmid, R; Müller, CE Discovery and Structure Relationships of Salicylanilide Derivatives as Potent, Non-acidic P2X1 Receptor Antagonists. J Med Chem63:6164-6178 (2020) [PubMed] Article
Tian, M; Abdelrahman, A; Baqi, Y; Fuentes, E; Azazna, D; Spanier, C; Densborn, S; Hinz, S; Schmid, R; Müller, CE Discovery and Structure Relationships of Salicylanilide Derivatives as Potent, Non-acidic P2X1 Receptor Antagonists. J Med Chem63:6164-6178 (2020) [PubMed] Article