

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Fusion glycoprotein F0 | ||
| Ligand | BDBM50542669 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_1987321 (CHEMBL4620868) | ||
| EC50 | 1.000000±n/a nM | ||
| Citation |  Vendeville, S; Tahri, A; Hu, L; Demin, S; Cooymans, L; Vos, A; Kwanten, L; Van den Berg, J; Battles, MB; McLellan, JS; Koul, A; Raboisson, P; Roymans, D; Jonckers, THM Discovery of 3-({5-Chloro-1-[3-(methylsulfonyl)propyl]-1 J Med Chem63:8046-8058 (2020) [PubMed] Article Vendeville, S; Tahri, A; Hu, L; Demin, S; Cooymans, L; Vos, A; Kwanten, L; Van den Berg, J; Battles, MB; McLellan, JS; Koul, A; Raboisson, P; Roymans, D; Jonckers, THM Discovery of 3-({5-Chloro-1-[3-(methylsulfonyl)propyl]-1 J Med Chem63:8046-8058 (2020) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Fusion glycoprotein F0 | |||
| Name: | Fusion glycoprotein F0 | ||
| Synonyms: | F | Fusion glycoprotein F0 | ||
| Type: | PROTEIN | ||
| Mol. Mass.: | 63485.30 | ||
| Organism: | Human respiratory syncytial virus | ||
| Description: | ChEMBL_106144 | ||
| Residue: | 574 | ||
| Sequence: |
| ||
| BDBM50542669 | |||
| n/a | |||
| Name | BDBM50542669 | ||
| Synonyms: | CHEMBL4646649 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C27H24ClN5O2 | ||
| Mol. Mass. | 485.965 | ||
| SMILES | COc1ccc(c(C)c1)-n1c2ccncc2n(Cc2cc3cc(Cl)ccc3n2CCCC#N)c1=O |(57.69,-18.88,;56.66,-20.03,;57.13,-21.49,;58.64,-21.82,;59.11,-23.28,;58.08,-24.42,;56.58,-24.11,;55.55,-25.25,;56.1,-22.64,;58.56,-25.88,;60.02,-26.36,;61.35,-25.59,;62.68,-26.35,;62.69,-27.9,;61.35,-28.67,;60.02,-27.9,;58.55,-28.38,;58.07,-29.84,;56.57,-30.16,;55.42,-29.12,;54.09,-29.89,;52.63,-29.42,;51.49,-30.44,;50.03,-29.97,;51.81,-31.95,;53.27,-32.42,;54.41,-31.4,;55.94,-31.56,;56.71,-32.9,;58.25,-32.9,;59.02,-34.23,;60.56,-34.24,;62.1,-34.23,;57.65,-27.13,;56.11,-27.13,)| | ||
| Structure | 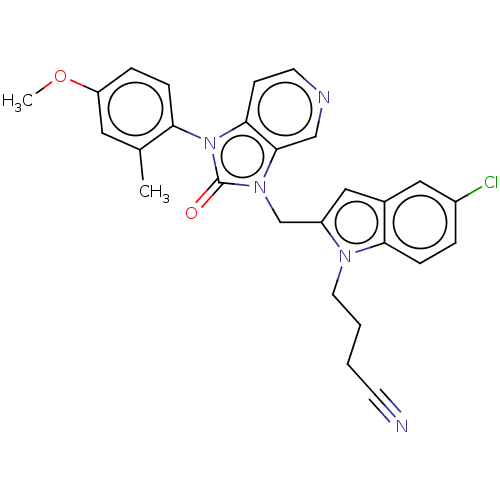
| ||