| Reaction Details |
|---|
 | Report a problem with these data |
| Target | N-lysine methyltransferase SMYD2 |
|---|
| Ligand | BDBM50550501 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2027562 (CHEMBL4681720) |
|---|
| IC50 | 8400±n/a nM |
|---|
| Citation |  Cowen, SD; Russell, D; Dakin, LA; Chen, H; Larsen, NA; Godin, R; Throner, S; Zheng, X; Molina, A; Wu, J; Cheung, T; Howard, T; Garcia-Arenas, R; Keen, N; Pendleton, CS; Pietenpol, JA; Ferguson, AD Design, Synthesis, and Biological Activity of Substrate Competitive SMYD2 Inhibitors. J Med Chem59:11079-11097 (2016) [PubMed] Cowen, SD; Russell, D; Dakin, LA; Chen, H; Larsen, NA; Godin, R; Throner, S; Zheng, X; Molina, A; Wu, J; Cheung, T; Howard, T; Garcia-Arenas, R; Keen, N; Pendleton, CS; Pietenpol, JA; Ferguson, AD Design, Synthesis, and Biological Activity of Substrate Competitive SMYD2 Inhibitors. J Med Chem59:11079-11097 (2016) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| N-lysine methyltransferase SMYD2 |
|---|
| Name: | N-lysine methyltransferase SMYD2 |
|---|
| Synonyms: | HSKM-B | Histone methyltransferase SMYD2 | KMT3C | Lysine N-methyltransferase 3C | N-lysine methyltransferase SMYD2 | SET and MYND domain-containing protein 2 | SET and MYND domain-containing protein 2 (SMYD2) | SMYD2 | SMYD2_HUMAN |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49689.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q9NRG4 |
|---|
| Residue: | 433 |
|---|
| Sequence: | MRAEGLGGLERFCSPGKGRGLRALQPFQVGDLLFSCPAYAYVLTVNERGNHCEYCFTRKE
GLSKCGRCKQAFYCNVECQKEDWPMHKLECSPMVVFGENWNPSETVRLTARILAKQKIHP
ERTPSEKLLAVKEFESHLDKLDNEKKDLIQSDIAALHHFYSKHLGFPDNDSLVVLFAQVN
CNGFTIEDEELSHLGSAIFPDVALMNHSCCPNVIVTYKGTLAEVRAVQEIKPGEEVFTSY
IDLLYPTEDRNDRLRDSYFFTCECQECTTKDKDKAKVEIRKLSDPPKAEAIRDMVRYARN
VIEEFRRAKHYKSPSELLEICELSQEKMSSVFEDSNVYMLHMMYQAMGVCLYMQDWEGAL
QYGQKIIKPYSKHYPLYSLNVASMWLKLGRLYMGLEHKAAGEKALKKAIAIMEVAHGKDH
PYISEIKQEIESH
|
|
|
|---|
| BDBM50550501 |
|---|
| n/a |
|---|
| Name | BDBM50550501 |
|---|
| Synonyms: | CHEMBL4751856 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H27N5O |
|---|
| Mol. Mass. | 377.4827 |
|---|
| SMILES | CCc1ccccc1-c1cc(C(=O)NCCCN2CCCC2)c2cn[nH]c2n1 |
|---|
| Structure | 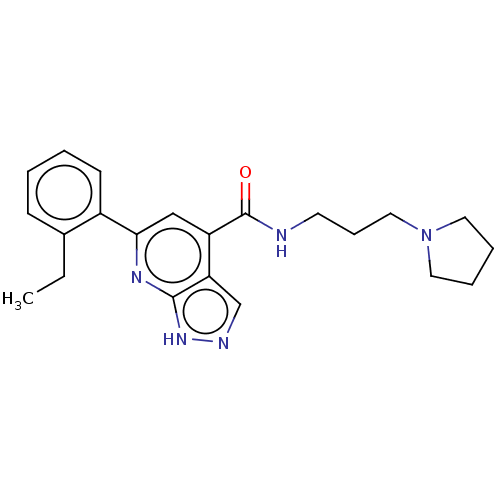
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cowen, SD; Russell, D; Dakin, LA; Chen, H; Larsen, NA; Godin, R; Throner, S; Zheng, X; Molina, A; Wu, J; Cheung, T; Howard, T; Garcia-Arenas, R; Keen, N; Pendleton, CS; Pietenpol, JA; Ferguson, AD Design, Synthesis, and Biological Activity of Substrate Competitive SMYD2 Inhibitors. J Med Chem59:11079-11097 (2016) [PubMed]
Cowen, SD; Russell, D; Dakin, LA; Chen, H; Larsen, NA; Godin, R; Throner, S; Zheng, X; Molina, A; Wu, J; Cheung, T; Howard, T; Garcia-Arenas, R; Keen, N; Pendleton, CS; Pietenpol, JA; Ferguson, AD Design, Synthesis, and Biological Activity of Substrate Competitive SMYD2 Inhibitors. J Med Chem59:11079-11097 (2016) [PubMed]