| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Genome polyprotein |
|---|
| Ligand | BDBM50111455 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_158823 (CHEMBL761632) |
|---|
| EC50 | 44±n/a nM |
|---|
| Citation |  Dragovich, PS; Prins, TJ; Zhou, R; Brown, EL; Maldonado, FC; Fuhrman, SA; Zalman, LS; Tuntland, T; Lee, CA; Patick, AK; Matthews, DA; Hendrickson, TF; Kosa, MB; Liu, B; Batugo, MR; Gleeson, JP; Sakata, SK; Chen, L; Guzman, MC; Meador, JW; Ferre, RA; Worland, ST Structure-based design, synthesis, and biological evaluation of irreversible human rhinovirus 3C protease inhibitors. 6. Structure-activity studies of orally bioavailable, 2-pyridone-containing peptidomimetics. J Med Chem45:1607-23 (2002) [PubMed] Dragovich, PS; Prins, TJ; Zhou, R; Brown, EL; Maldonado, FC; Fuhrman, SA; Zalman, LS; Tuntland, T; Lee, CA; Patick, AK; Matthews, DA; Hendrickson, TF; Kosa, MB; Liu, B; Batugo, MR; Gleeson, JP; Sakata, SK; Chen, L; Guzman, MC; Meador, JW; Ferre, RA; Worland, ST Structure-based design, synthesis, and biological evaluation of irreversible human rhinovirus 3C protease inhibitors. 6. Structure-activity studies of orally bioavailable, 2-pyridone-containing peptidomimetics. J Med Chem45:1607-23 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Genome polyprotein |
|---|
| Name: | Genome polyprotein |
|---|
| Synonyms: | Human rhinovirus A protease | Human rhinovirus B 3A protease |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 44361.04 |
|---|
| Organism: | Human rhinovirus B |
|---|
| Description: | ChEMBL_158953 |
|---|
| Residue: | 401 |
|---|
| Sequence: | AFRPCNVNTKIGNAKCCPFVCGKAVTFKDRSTCSTYNLSSSLHHILEEDKRRRQVVDVMS
AIFQGPISLDAPPPPAIADLLQSVRTPRVIKYCQIIMGHPAECQVERDLNIANSIIAIIA
NIISIAGIIFVIYKLFCSLQGPYSGEPKPKTKVPERRVVAQGPEEEFGRSILKNNTCVIT
TGNGKFTGLGIHDRILIIPTHADPGREVQVNGVHTKVLDSYDLYNRDGVKLEITVIQLDR
NEKFRDIRKYIPETEDDYPECNLALSANQDEPTIIKVGDVVSYGNILLSGNQTARMLKYN
YPTKSGYCGGVLYKIGQILGIHVGGNGRDGFSAMLLRSYFTGQIKVNKHATECGLPDIQT
IHTPSKTKLQPSVFYDVFPGSKEPAVLTDNDPRLEVNFKEA
|
|
|
|---|
| BDBM50111455 |
|---|
| n/a |
|---|
| Name | BDBM50111455 |
|---|
| Synonyms: | (1S,5S)-4-((S)-3-(3,4-Difluoro-phenyl)-2-{3-[(5-methyl-isoxazole-3-carbonyl)-amino]-2-oxo-2H-pyridin-1-yl}-propionylamino)-5-(2-oxo-pyrrolidin-3-yl)-pent-2-enoic acid isopropyl ester | 4-(3-(3,4-Difluoro-phenyl)-2-{3-[(5-methyl-isoxazole-3-carbonyl)-amino]-2-oxo-2H-pyridin-1-yl}-propionylamino)-5-(2-oxo-pyrrolidin-3-yl)-pent-2-enoic acid isopropyl ester | CHEMBL291276 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H33F2N5O7 |
|---|
| Mol. Mass. | 625.6198 |
|---|
| SMILES | CC(C)OC(=O)\C=C\[C@H](C[C@@H]1CCNC1=O)NC(=O)[C@H](Cc1ccc(F)c(F)c1)n1cccc(NC(=O)c2cc(C)on2)c1=O |
|---|
| Structure | 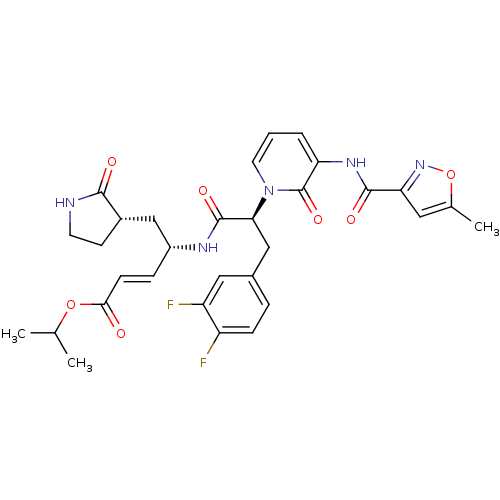
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dragovich, PS; Prins, TJ; Zhou, R; Brown, EL; Maldonado, FC; Fuhrman, SA; Zalman, LS; Tuntland, T; Lee, CA; Patick, AK; Matthews, DA; Hendrickson, TF; Kosa, MB; Liu, B; Batugo, MR; Gleeson, JP; Sakata, SK; Chen, L; Guzman, MC; Meador, JW; Ferre, RA; Worland, ST Structure-based design, synthesis, and biological evaluation of irreversible human rhinovirus 3C protease inhibitors. 6. Structure-activity studies of orally bioavailable, 2-pyridone-containing peptidomimetics. J Med Chem45:1607-23 (2002) [PubMed]
Dragovich, PS; Prins, TJ; Zhou, R; Brown, EL; Maldonado, FC; Fuhrman, SA; Zalman, LS; Tuntland, T; Lee, CA; Patick, AK; Matthews, DA; Hendrickson, TF; Kosa, MB; Liu, B; Batugo, MR; Gleeson, JP; Sakata, SK; Chen, L; Guzman, MC; Meador, JW; Ferre, RA; Worland, ST Structure-based design, synthesis, and biological evaluation of irreversible human rhinovirus 3C protease inhibitors. 6. Structure-activity studies of orally bioavailable, 2-pyridone-containing peptidomimetics. J Med Chem45:1607-23 (2002) [PubMed]