| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Ribosyldihydronicotinamide dehydrogenase [quinone] |
|---|
| Ligand | BDBM50112204 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_104942 (CHEMBL713109) |
|---|
| Ki | 70±n/a nM |
|---|
| Citation |  Leclerc, V; Yous, S; Delagrange, P; Boutin, JA; Renard, P; Lesieur, D Synthesis of nitroindole derivatives with high affinity and selectivity for melatoninergic binding sites MT(3). J Med Chem45:1853-9 (2002) [PubMed] Leclerc, V; Yous, S; Delagrange, P; Boutin, JA; Renard, P; Lesieur, D Synthesis of nitroindole derivatives with high affinity and selectivity for melatoninergic binding sites MT(3). J Med Chem45:1853-9 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Ribosyldihydronicotinamide dehydrogenase [quinone] |
|---|
| Name: | Ribosyldihydronicotinamide dehydrogenase [quinone] |
|---|
| Synonyms: | Metallothionein-3 | NMOR2 | NQO2 | NQO2_HUMAN | NRH dehydrogenase [quinone] 2 | NRH:quinone oxidoreductase 2 | QR2 | Quinone reductase 2 | Quinone reductase 2 (NQO2) | Ribosyldihydronicotinamide dehydrogenase [quinone] |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 25917.25 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P16083 |
|---|
| Residue: | 231 |
|---|
| Sequence: | MAGKKVLIVYAHQEPKSFNGSLKNVAVDELSRQGCTVTVSDLYAMNLEPRATDKDITGTL
SNPEVFNYGVETHEAYKQRSLASDITDEQKKVREADLVIFQFPLYWFSVPAILKGWMDRV
LCQGFAFDIPGFYDSGLLQGKLALLSVTTGGTAEMYTKTGVNGDSRYFLWPLQHGTLHFC
GFKVLAPQISFAPEIASEEERKGMVAAWSQRLQTIWKEEPIPCTAHWHFGQ
|
|
|
|---|
| BDBM50112204 |
|---|
| n/a |
|---|
| Name | BDBM50112204 |
|---|
| Synonyms: | CHEMBL52755 | N-[2-(2-Iodo-5-methoxy-6-nitro-1H-indol-3-yl)-ethyl]-acetamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H14IN3O4 |
|---|
| Mol. Mass. | 403.1724 |
|---|
| SMILES | COc1cc2c(CCNC(C)=O)c(I)[nH]c2cc1[N+]([O-])=O |
|---|
| Structure | 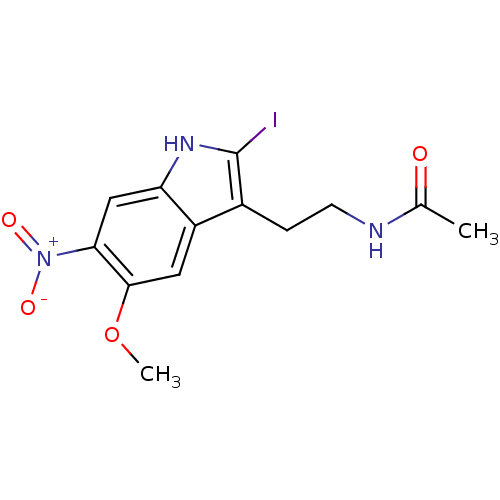
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Leclerc, V; Yous, S; Delagrange, P; Boutin, JA; Renard, P; Lesieur, D Synthesis of nitroindole derivatives with high affinity and selectivity for melatoninergic binding sites MT(3). J Med Chem45:1853-9 (2002) [PubMed]
Leclerc, V; Yous, S; Delagrange, P; Boutin, JA; Renard, P; Lesieur, D Synthesis of nitroindole derivatives with high affinity and selectivity for melatoninergic binding sites MT(3). J Med Chem45:1853-9 (2002) [PubMed]