| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50112524 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_208903 |
|---|
| Ki | 3000±n/a nM |
|---|
| Citation |  Shaw, KJ; Guilford, WJ; Griedel, BD; Sakata, S; Trinh, L; Wu, S; Xu, W; Zhao, Z; Morrissey, MM Benzimidazole-based fXa inhibitors with improved thrombin and trypsin selectivity. Bioorg Med Chem Lett12:1311-4 (2002) [PubMed] Shaw, KJ; Guilford, WJ; Griedel, BD; Sakata, S; Trinh, L; Wu, S; Xu, W; Zhao, Z; Morrissey, MM Benzimidazole-based fXa inhibitors with improved thrombin and trypsin selectivity. Bioorg Med Chem Lett12:1311-4 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50112524 |
|---|
| n/a |
|---|
| Name | BDBM50112524 |
|---|
| Synonyms: | 7-{2-Ethyl-5-[1-(1-imino-ethyl)-piperidin-4-yloxy]-4-nitro-benzoimidazol-1-ylmethyl}-naphthalene-2-carboxamidine | CHEMBL25073 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31N7O3 |
|---|
| Mol. Mass. | 513.5908 |
|---|
| SMILES | n/a |
|---|
| Structure | 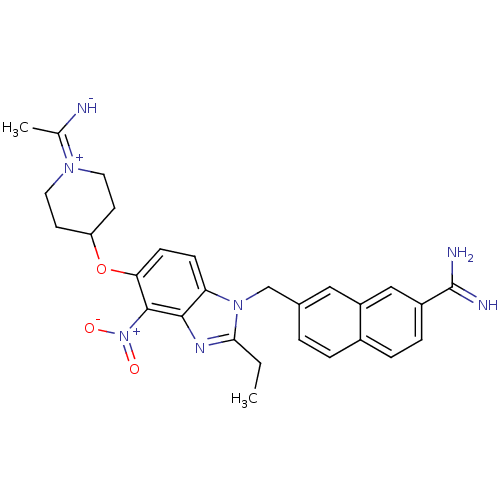
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shaw, KJ; Guilford, WJ; Griedel, BD; Sakata, S; Trinh, L; Wu, S; Xu, W; Zhao, Z; Morrissey, MM Benzimidazole-based fXa inhibitors with improved thrombin and trypsin selectivity. Bioorg Med Chem Lett12:1311-4 (2002) [PubMed]
Shaw, KJ; Guilford, WJ; Griedel, BD; Sakata, S; Trinh, L; Wu, S; Xu, W; Zhao, Z; Morrissey, MM Benzimidazole-based fXa inhibitors with improved thrombin and trypsin selectivity. Bioorg Med Chem Lett12:1311-4 (2002) [PubMed]