| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cathepsin D |
|---|
| Ligand | BDBM50080960 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2051501 (CHEMBL4706200) |
|---|
| Ki | 0.700000±n/a nM |
|---|
| Citation |  Goyal, S; Patel, KV; Nagare, Y; Raykar, DB; Raikar, SS; Dolas, A; Khurana, P; Cyriac, R; Sarak, S; Gangar, M; Agarwal, AK; Kulkarni, A Identification and structure-activity relationship studies of small molecule inhibitors of the human cathepsin D. Bioorg Med Chem29:0 (2021) [PubMed] Article Goyal, S; Patel, KV; Nagare, Y; Raykar, DB; Raikar, SS; Dolas, A; Khurana, P; Cyriac, R; Sarak, S; Gangar, M; Agarwal, AK; Kulkarni, A Identification and structure-activity relationship studies of small molecule inhibitors of the human cathepsin D. Bioorg Med Chem29:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cathepsin D |
|---|
| Name: | Cathepsin D |
|---|
| Synonyms: | CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 44551.72 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated. |
|---|
| Residue: | 412 |
|---|
| Sequence: | MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVP
AVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIH
HKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFG
EATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQ
PGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSL
MVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQ
AGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
|
|
|
|---|
| BDBM50080960 |
|---|
| n/a |
|---|
| Name | BDBM50080960 |
|---|
| Synonyms: | 2-Bromo-N-[(1S,2S)-3-{[2-(2,4-dichloro-phenyl)-ethyl]-[3-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-propionyl]-amino}-2-hydroxy-1-(3-phenoxy-benzyl)-propyl]-4,5-dimethoxy-benzamide | 2-Bromo-N-[(1S,3S)-3-{[2-(2,4-dichloro-phenyl)-ethyl]-[3-(1,3-dioxo-1,3-dihydro-isoindol-2-yl)-propionyl]-amino}-2-hydroxy-1-(3-phenoxy-benzyl)-propyl]-4,5-dimethoxy-benzamide | CHEMBL81927 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C44H40BrCl2N3O8 |
|---|
| Mol. Mass. | 889.614 |
|---|
| SMILES | COc1cc(Br)c(cc1OC)C(=O)N[C@@H](Cc1cccc(Oc2ccccc2)c1)[C@@H](O)CN(CCc1ccc(Cl)cc1Cl)C(=O)CCN1C(=O)c2ccccc2C1=O |
|---|
| Structure | 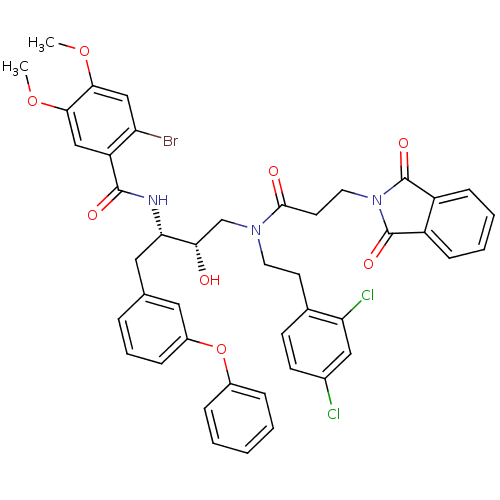
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Goyal, S; Patel, KV; Nagare, Y; Raykar, DB; Raikar, SS; Dolas, A; Khurana, P; Cyriac, R; Sarak, S; Gangar, M; Agarwal, AK; Kulkarni, A Identification and structure-activity relationship studies of small molecule inhibitors of the human cathepsin D. Bioorg Med Chem29:0 (2021) [PubMed] Article
Goyal, S; Patel, KV; Nagare, Y; Raykar, DB; Raikar, SS; Dolas, A; Khurana, P; Cyriac, R; Sarak, S; Gangar, M; Agarwal, AK; Kulkarni, A Identification and structure-activity relationship studies of small molecule inhibitors of the human cathepsin D. Bioorg Med Chem29:0 (2021) [PubMed] Article