| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50555233 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2051621 (CHEMBL4706320) |
|---|
| IC50 | >50000±n/a nM |
|---|
| Citation |  Davies, DT; Leiris, S; Zalacain, M; Sprynski, N; Castandet, J; Bousquet, J; Lozano, C; Llanos, A; Alibaud, L; Vasa, S; Pattipati, R; Valige, R; Kummari, B; Pothukanuri, S; De Piano, C; Morrissey, I; Holden, K; Warn, P; Marcoccia, F; Benvenuti, M; Pozzi, C; Tassone, G; Mangani, S; Docquier, JD; Pallin, D; Elliot, R; Lemonnier, M; Everett, M Discovery of J Med Chem63:15802-15820 (2020) [PubMed] Article Davies, DT; Leiris, S; Zalacain, M; Sprynski, N; Castandet, J; Bousquet, J; Lozano, C; Llanos, A; Alibaud, L; Vasa, S; Pattipati, R; Valige, R; Kummari, B; Pothukanuri, S; De Piano, C; Morrissey, I; Holden, K; Warn, P; Marcoccia, F; Benvenuti, M; Pozzi, C; Tassone, G; Mangani, S; Docquier, JD; Pallin, D; Elliot, R; Lemonnier, M; Everett, M Discovery of J Med Chem63:15802-15820 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50555233 |
|---|
| n/a |
|---|
| Name | BDBM50555233 |
|---|
| Synonyms: | CHEMBL4753698 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C6H8FN2NaO5S |
|---|
| Mol. Mass. | 262.191 |
|---|
| SMILES | [Na;v0+].[H][C@]12[#6]-[#7](-[#6@H](F)-[#6]-[#6]1)-[#6](=O)-[#7]2-[#8]S([#8-])(=O)=O |r| |
|---|
| Structure | 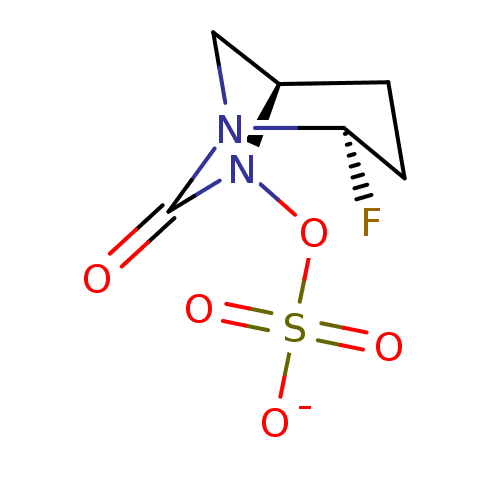
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Davies, DT; Leiris, S; Zalacain, M; Sprynski, N; Castandet, J; Bousquet, J; Lozano, C; Llanos, A; Alibaud, L; Vasa, S; Pattipati, R; Valige, R; Kummari, B; Pothukanuri, S; De Piano, C; Morrissey, I; Holden, K; Warn, P; Marcoccia, F; Benvenuti, M; Pozzi, C; Tassone, G; Mangani, S; Docquier, JD; Pallin, D; Elliot, R; Lemonnier, M; Everett, M Discovery of J Med Chem63:15802-15820 (2020) [PubMed] Article
Davies, DT; Leiris, S; Zalacain, M; Sprynski, N; Castandet, J; Bousquet, J; Lozano, C; Llanos, A; Alibaud, L; Vasa, S; Pattipati, R; Valige, R; Kummari, B; Pothukanuri, S; De Piano, C; Morrissey, I; Holden, K; Warn, P; Marcoccia, F; Benvenuti, M; Pozzi, C; Tassone, G; Mangani, S; Docquier, JD; Pallin, D; Elliot, R; Lemonnier, M; Everett, M Discovery of J Med Chem63:15802-15820 (2020) [PubMed] Article