| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Endothelin-1 receptor |
|---|
| Ligand | BDBM50117909 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_65473 (CHEMBL876873) |
|---|
| IC50 | 24±n/a nM |
|---|
| Citation |  Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed] Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Endothelin-1 receptor |
|---|
| Name: | Endothelin-1 receptor |
|---|
| Synonyms: | EDNRA | EDNRA_HUMAN | ET-A | ETA | ETA-R | ETRA | Endothelin receptor type A | Endothelin receptor, ET-A/ET-B | hET-AR |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 48736.88 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25101 |
|---|
| Residue: | 427 |
|---|
| Sequence: | METLCLRASFWLALVGCVISDNPERYSTNLSNHVDDFTTFRGTELSFLVTTHQPTNLVLP
SNGSMHNYCPQQTKITSAFKYINTVISCTIFIVGMVGNATLLRIIYQNKCMRNGPNALIA
SLALGDLIYVVIDLPINVFKLLAGRWPFDHNDFGVFLCKLFPFLQKSSVGITVLNLCALS
VDRYRAVASWSRVQGIGIPLVTAIEIVSIWILSFILAIPEAIGFVMVPFEYRGEQHKTCM
LNATSKFMEFYQDVKDWWLFGFYFCMPLVCTAIFYTLMTCEMLNRRNGSLRIALSEHLKQ
RREVAKTVFCLVVIFALCWFPLHLSRILKKTVYNEMDKNRCELLSFLLLMDYIGINLATM
NSCINPIALYFVSKKFKNCFQSCLCCCCYQSKSLMTSVPMNGTSIQWKNHDQNNHNTDRS
SHKDSMN
|
|
|
|---|
| BDBM50117909 |
|---|
| n/a |
|---|
| Name | BDBM50117909 |
|---|
| Synonyms: | CHEMBL278176 | N-{2-Benzo[1,3]dioxol-5-yl-2-[4-(2-ethyl-5,7-dimethyl-imidazo[4,5-b]pyridin-3-ylmethyl)-2-propyl-phenoxy]-acetyl}-4-isopropyl-benzenesulfonamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C38H42N4O6S |
|---|
| Mol. Mass. | 682.828 |
|---|
| SMILES | CCCc1cc(Cn2c(CC)nc3c(C)cc(C)nc23)ccc1OC(C(=O)NS(=O)(=O)c1ccc(cc1)C(C)C)c1ccc2OCOc2c1 |
|---|
| Structure | 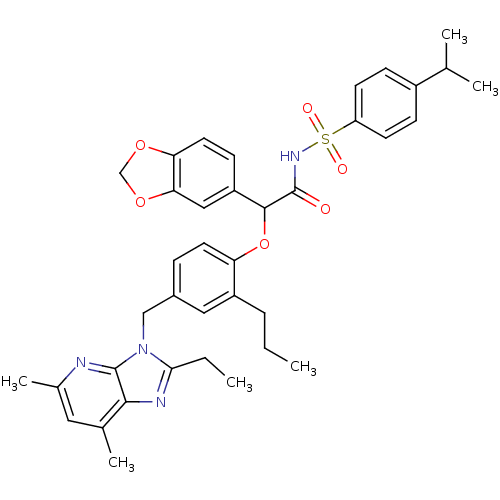
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed]
Murugesan, N; Tellew, JE; Gu, Z; Kunst, BL; Fadnis, L; Cornelius, LA; Baska, RA; Yang, Y; Beyer, SM; Monshizadegan, H; Dickinson, KE; Panchal, B; Valentine, MT; Chong, S; Morrison, RA; Carlson, KE; Powell, JR; Moreland, S; Barrish, JC; Kowala, MC; Macor, JE Discovery of N-isoxazolyl biphenylsulfonamides as potent dual angiotensin II and endothelin A receptor antagonists. J Med Chem45:3829-35 (2002) [PubMed]