| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50118705 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34199 (CHEMBL649216) |
|---|
| Ki | 880.0±n/a nM |
|---|
| Citation |  Altenbach, RJ; Khilevich, A; Meyer, MD; Buckner, SA; Milicic, I; Daza, AV; Brune, ME; O'Neill, AB; Gauvin, DM; Cain, JC; Nakane, M; Holladay, MW; Williams, M; Brioni, JD; Sullivan, JP N-[3-(1H-imidazol-4-ylmethyl)phenyl]ethanesulfonamide (ABT-866, 1),(1) a novel alpha(1)-adrenoceptor ligand with an enhanced in vitro and in vivo profile relative to phenylpropanolamine and midodrine. J Med Chem45:4395-7 (2002) [PubMed] Altenbach, RJ; Khilevich, A; Meyer, MD; Buckner, SA; Milicic, I; Daza, AV; Brune, ME; O'Neill, AB; Gauvin, DM; Cain, JC; Nakane, M; Holladay, MW; Williams, M; Brioni, JD; Sullivan, JP N-[3-(1H-imidazol-4-ylmethyl)phenyl]ethanesulfonamide (ABT-866, 1),(1) a novel alpha(1)-adrenoceptor ligand with an enhanced in vitro and in vivo profile relative to phenylpropanolamine and midodrine. J Med Chem45:4395-7 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_HUMAN | ADRA1B | Adrenergic alpha1B | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha-1B adrenergic receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56862.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35368 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKNANFTGPNQTSSNSTLPQLDITRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAMADLLLSFTVLPFSAALEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFV
RILGCQCRGRGRRRRRRRRRLGGCAYTYRPWTRGGSLERSQSRKDSLDDSGSCLSGSQRT
LPSASPSPGYLGRGAPPPVELCAFPEWKAPGALLSLPAPEPPGRRGRHDSGPLFTFKLLT
EPESPGTDGGASNGGCEAAADVANGQPGFKSNMPLAPGQF
|
|
|
|---|
| BDBM50118705 |
|---|
| n/a |
|---|
| Name | BDBM50118705 |
|---|
| Synonyms: | ABT-866 | CHEMBL326702 | Ethanesulfonic acid [3-(1H-imidazol-4-ylmethyl)-phenyl]-amide | N-(3-((1H-imidazol-4-yl)methyl)phenyl)ethanesulfonamide | N-[3-(1H-imidazol-4-ylmethyl)-phenyl]ethanesulfonamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H15N3O2S |
|---|
| Mol. Mass. | 265.331 |
|---|
| SMILES | CCS(=O)(=O)Nc1cccc(Cc2cnc[nH]2)c1 |
|---|
| Structure | 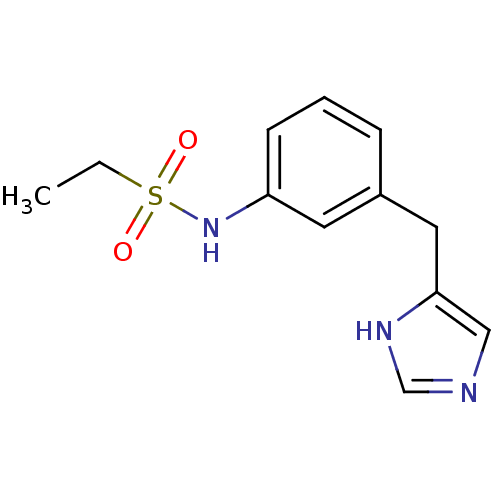
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Altenbach, RJ; Khilevich, A; Meyer, MD; Buckner, SA; Milicic, I; Daza, AV; Brune, ME; O'Neill, AB; Gauvin, DM; Cain, JC; Nakane, M; Holladay, MW; Williams, M; Brioni, JD; Sullivan, JP N-[3-(1H-imidazol-4-ylmethyl)phenyl]ethanesulfonamide (ABT-866, 1),(1) a novel alpha(1)-adrenoceptor ligand with an enhanced in vitro and in vivo profile relative to phenylpropanolamine and midodrine. J Med Chem45:4395-7 (2002) [PubMed]
Altenbach, RJ; Khilevich, A; Meyer, MD; Buckner, SA; Milicic, I; Daza, AV; Brune, ME; O'Neill, AB; Gauvin, DM; Cain, JC; Nakane, M; Holladay, MW; Williams, M; Brioni, JD; Sullivan, JP N-[3-(1H-imidazol-4-ylmethyl)phenyl]ethanesulfonamide (ABT-866, 1),(1) a novel alpha(1)-adrenoceptor ligand with an enhanced in vitro and in vivo profile relative to phenylpropanolamine and midodrine. J Med Chem45:4395-7 (2002) [PubMed]