| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50170678 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2064601 (CHEMBL4719854) |
|---|
| EC50 | 3400±n/a nM |
|---|
| Citation |  Crowley, RS; Riley, AP; Sherwood, AM; Groer, CE; Shivaperumal, N; Biscaia, M; Paton, K; Schneider, S; Provasi, D; Kivell, BM; Filizola, M; Prisinzano, TE Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Identification of a Potent and Centrally Acting ? Opioid Analgesic with Reduced Abuse Liability. J Med Chem59:11027-11038 (2016) [PubMed] Article Crowley, RS; Riley, AP; Sherwood, AM; Groer, CE; Shivaperumal, N; Biscaia, M; Paton, K; Schneider, S; Provasi, D; Kivell, BM; Filizola, M; Prisinzano, TE Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Identification of a Potent and Centrally Acting ? Opioid Analgesic with Reduced Abuse Liability. J Med Chem59:11027-11038 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | M-OR-1 | MOP | MOR-1 | MOR1 | MUOR1 | Mu Opioid Receptor | Mu opiate receptor | OPIATE Mu | OPRM1 | OPRM_HUMAN | hMOP | mu-type opioid receptor isoform MOR-1 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44789.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35372 |
|---|
| Residue: | 400 |
|---|
| Sequence: | MDSSAAPTNASNCTDALAYSSCSPAPSPGSWVNLSHLDGNLSDPCGPNRTDLGGRDSLCP
PTGSPSMITAITIMALYSIVCVVGLFGNFLVMYVIVRYTKMKTATNIYIFNLALADALAT
STLPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSIFTLCTMSVDRYIAVCHPVKALDF
RTPRNAKIINVCNWILSSAIGLPVMFMATTKYRQGSIDCTLTFSHPTWYWENLLKICVFI
FAFIMPVLIITVCYGLMILRLKSVRMLSGSKEKDRNLRRITRMVLVVVAVFIVCWTPIHI
YVIIKALVTIPETTFQTVSWHFCIALGYTNSCLNPVLYAFLDENFKRCFREFCIPTSSNI
EQQNSTRIRQNTRDHPSTANTVDRTNHQLENLEAETAPLP
|
|
|
|---|
| BDBM50170678 |
|---|
| n/a |
|---|
| Name | BDBM50170678 |
|---|
| Synonyms: | (3S,4aR,4bS,6S,8R,8aR,10aR)-6-Benzoyloxy-3-furan-3-yl-4a,8a-dimethyl-1,5-dioxo-dodecahydro-2-oxa-phenanthrene-8-carboxylic acid methyl ester | CHEMBL363324 | HERKINORIN |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H30O8 |
|---|
| Mol. Mass. | 494.533 |
|---|
| SMILES | COC(=O)[C@@H]1C[C@H](OC(=O)c2ccccc2)C(=O)[C@H]2[C@@]1(C)CC[C@H]1C(=O)O[C@@H](C[C@]21C)c1ccoc1 |r| |
|---|
| Structure | 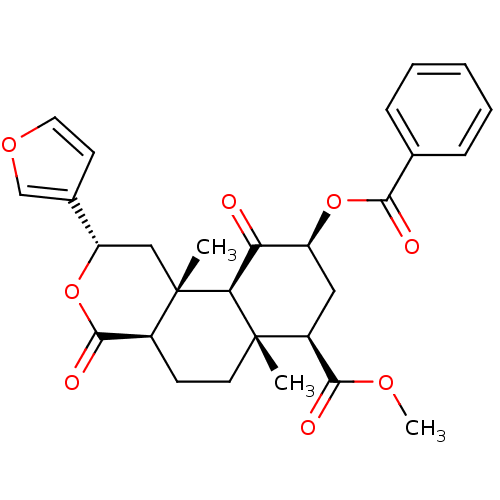
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Crowley, RS; Riley, AP; Sherwood, AM; Groer, CE; Shivaperumal, N; Biscaia, M; Paton, K; Schneider, S; Provasi, D; Kivell, BM; Filizola, M; Prisinzano, TE Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Identification of a Potent and Centrally Acting ? Opioid Analgesic with Reduced Abuse Liability. J Med Chem59:11027-11038 (2016) [PubMed] Article
Crowley, RS; Riley, AP; Sherwood, AM; Groer, CE; Shivaperumal, N; Biscaia, M; Paton, K; Schneider, S; Provasi, D; Kivell, BM; Filizola, M; Prisinzano, TE Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Identification of a Potent and Centrally Acting ? Opioid Analgesic with Reduced Abuse Liability. J Med Chem59:11027-11038 (2016) [PubMed] Article