| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Ligand | BDBM50127149 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_53333 (CHEMBL664919) |
|---|
| IC50 | 1600±n/a nM |
|---|
| Citation |  Rosowsky, A; Forsch, RA; Queener, SF Further studies on 2,4-diamino-5-(2',5'-disubstituted benzyl)pyrimidines as potent and selective inhibitors of dihydrofolate reductases from three major opportunistic pathogens of AIDS. J Med Chem46:1726-36 (2003) [PubMed] Article Rosowsky, A; Forsch, RA; Queener, SF Further studies on 2,4-diamino-5-(2',5'-disubstituted benzyl)pyrimidines as potent and selective inhibitors of dihydrofolate reductases from three major opportunistic pathogens of AIDS. J Med Chem46:1726-36 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Name: | Bifunctional dihydrofolate reductase-thymidylate synthase |
|---|
| Synonyms: | DHFR-TS | DRTS_TOXGO | Dihydrofolate reductase | Dihydrofolate reductase (DHFR) | Dihydrofolate reductase; T. gondii vs rat |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68757.49 |
|---|
| Organism: | Toxoplasma gondii |
|---|
| Description: | Q07422 |
|---|
| Residue: | 610 |
|---|
| Sequence: | MQKPVCLVVAMTPKRGIGINNGLPWPHLTTDFKHFSRVTKTTPEEASRLNGWLPRKFAKT
GDSGLPSPSVGKRFNAVVMGRKTWESMPRKFRPLVDRLNIVVSSSLKEEDIAAEKPQAEG
QQRVRVCASLPAALSLLEEEYKDSVDQIFVVGGAGLYEAALSLGVASHLYITRVAREFPC
DVFFPAFPGDDILSNKSTAAQAAAPAESVFVPFCPELGREKDNEATYRPIFISKTFSDNG
VPYDFVVLEKRRKTDDAATAEPSNAMSSLTSTRETTPVHGLQAPSSAAAIAPVLAWMDEE
DRKKREQKELIRAVPHVHFRGHEEFQYLDLIADIINNGRTMDDRTGVGVISKFGCTMRYS
LDQAFPLLTTKRVFWKGVLEELLWFIRGDTNANHLSEKGVKIWDKNVTREFLDSRNLPHR
EVGDIGPGYGFQWRHFGAAYKDMHTDYTGQGVDQLKNVIQMLRTNPTDRRMLMTAWNPAA
LDEMALPPCHLLCQFYVNDQKELSCIMYQRSCDVGLGVPFNIASYSLLTLMVAHVCNLKP
KEFIHFMGNTHVYTNHVEALKEQLRREPRPFPIVNILNKERIKEIDDFTAEDFEVVGYVP
HGRIQMEMAV
|
|
|
|---|
| BDBM50127149 |
|---|
| n/a |
|---|
| Name | BDBM50127149 |
|---|
| Synonyms: | 3-{3-[3-(2,4-Diamino-pyrimidin-5-ylmethyl)-4-methoxy-phenyl]-prop-2-ynyloxy}-benzoic acid | CHEMBL35080 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H20N4O4 |
|---|
| Mol. Mass. | 404.4186 |
|---|
| SMILES | COc1ccc(cc1Cc1cnc(N)nc1N)C#CCOc1cccc(c1)C(O)=O |
|---|
| Structure | 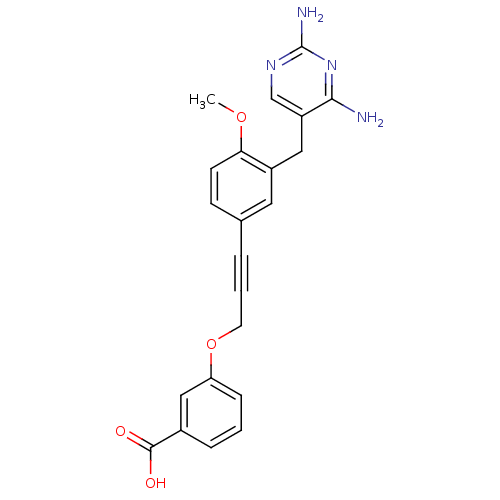
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Rosowsky, A; Forsch, RA; Queener, SF Further studies on 2,4-diamino-5-(2',5'-disubstituted benzyl)pyrimidines as potent and selective inhibitors of dihydrofolate reductases from three major opportunistic pathogens of AIDS. J Med Chem46:1726-36 (2003) [PubMed] Article
Rosowsky, A; Forsch, RA; Queener, SF Further studies on 2,4-diamino-5-(2',5'-disubstituted benzyl)pyrimidines as potent and selective inhibitors of dihydrofolate reductases from three major opportunistic pathogens of AIDS. J Med Chem46:1726-36 (2003) [PubMed] Article