| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cystic fibrosis transmembrane conductance regulator |
|---|
| Ligand | BDBM50032693 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2086891 (CHEMBL4768154) |
|---|
| EC50 | 12±n/a nM |
|---|
| Citation |  Froux, L; Elbahnsi, A; Boucherle, B; Billet, A; Baatallah, N; Hoffmann, B; Alliot, J; Zelli, R; Zeinyeh, W; Haudecoeur, R; Chevalier, B; Fortun�, A; Mirval, S; Simard, C; Lehn, P; Mornon, JP; Hinzpeter, A; Becq, F; Callebaut, I; D�cout, JL Targeting different binding sites in the CFTR structures allows to synergistically potentiate channel activity. Eur J Med Chem190:0 (2020) [PubMed] Article Froux, L; Elbahnsi, A; Boucherle, B; Billet, A; Baatallah, N; Hoffmann, B; Alliot, J; Zelli, R; Zeinyeh, W; Haudecoeur, R; Chevalier, B; Fortun�, A; Mirval, S; Simard, C; Lehn, P; Mornon, JP; Hinzpeter, A; Becq, F; Callebaut, I; D�cout, JL Targeting different binding sites in the CFTR structures allows to synergistically potentiate channel activity. Eur J Med Chem190:0 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cystic fibrosis transmembrane conductance regulator |
|---|
| Name: | Cystic fibrosis transmembrane conductance regulator |
|---|
| Synonyms: | ABCC7 | ATP-binding cassette sub-family C member 7 | CFTR | CFTR_HUMAN | Channel conductance-controlling ATPase | cAMP-dependent chloride channel |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 168171.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_90421313 |
|---|
| Residue: | 1480 |
|---|
| Sequence: | MQRSPLEKASVVSKLFFSWTRPILRKGYRQRLELSDIYQIPSVDSADNLSEKLEREWDRE
LASKKNPKLINALRRCFFWRFMFYGIFLYLGEVTKAVQPLLLGRIIASYDPDNKEERSIA
IYLGIGLCLLFIVRTLLLHPAIFGLHHIGMQMRIAMFSLIYKKTLKLSSRVLDKISIGQL
VSLLSNNLNKFDEGLALAHFVWIAPLQVALLMGLIWELLQASAFCGLGFLIVLALFQAGL
GRMMMKYRDQRAGKISERLVITSEMIENIQSVKAYCWEEAMEKMIENLRQTELKLTRKAA
YVRYFNSSAFFFSGFFVVFLSVLPYALIKGIILRKIFTTISFCIVLRMAVTRQFPWAVQT
WYDSLGAINKIQDFLQKQEYKTLEYNLTTTEVVMENVTAFWEEGFGELFEKAKQNNNNRK
TSNGDDSLFFSNFSLLGTPVLKDINFKIERGQLLAVAGSTGAGKTSLLMVIMGELEPSEG
KIKHSGRISFCSQFSWIMPGTIKENIIFGVSYDEYRYRSVIKACQLEEDISKFAEKDNIV
LGEGGITLSGGQRARISLARAVYKDADLYLLDSPFGYLDVLTEKEIFESCVCKLMANKTR
ILVTSKMEHLKKADKILILHEGSSYFYGTFSELQNLQPDFSSKLMGCDSFDQFSAERRNS
ILTETLHRFSLEGDAPVSWTETKKQSFKQTGEFGEKRKNSILNPINSIRKFSIVQKTPLQ
MNGIEEDSDEPLERRLSLVPDSEQGEAILPRISVISTGPTLQARRRQSVLNLMTHSVNQG
QNIHRKTTASTRKVSLAPQANLTELDIYSRRLSQETGLEISEEINEEDLKECFFDDMESI
PAVTTWNTYLRYITVHKSLIFVLIWCLVIFLAEVAASLVVLWLLGNTPLQDKGNSTHSRN
NSYAVIITSTSSYYVFYIYVGVADTLLAMGFFRGLPLVHTLITVSKILHHKMLHSVLQAP
MSTLNTLKAGGILNRFSKDIAILDDLLPLTIFDFIQLLLIVIGAIAVVAVLQPYIFVATV
PVIVAFIMLRAYFLQTSQQLKQLESEGRSPIFTHLVTSLKGLWTLRAFGRQPYFETLFHK
ALNLHTANWFLYLSTLRWFQMRIEMIFVIFFIAVTFISILTTGEGEGRVGIILTLAMNIM
STLQWAVNSSIDVDSLMRSVSRVFKFIDMPTEGKPTKSTKPYKNGQLSKVMIIENSHVKK
DDIWPSGGQMTVKDLTAKYTEGGNAILENISFSISPGQRVGLLGRTGSGKSTLLSAFLRL
LNTEGEIQIDGVSWDSITLQQWRKAFGVIPQKVFIFSGTFRKNLDPYEQWSDQEIWKVAD
EVGLRSVIEQFPGKLDFVLVDGGCVLSHGHKQLMCLARSVLSKAKILLLDEPSAHLDPVT
YQIIRRTLKQAFADCTVILCEHRIEAMLECQQFLVIEENKVRQYDSIQKLLNERSLFRQA
ISPSDRVKLFPHRNSSKCKSKPQIAALKEETEEEVQDTRL
|
|
|
|---|
| BDBM50032693 |
|---|
| n/a |
|---|
| Name | BDBM50032693 |
|---|
| Synonyms: | CHEBI:66901 | Ivacaftor | Kalydeco | VX-770 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H28N2O3 |
|---|
| Mol. Mass. | 392.4907 |
|---|
| SMILES | CC(C)(C)c1cc(c(NC(=O)c2c[nH]c3ccccc3c2=O)cc1O)C(C)(C)C |
|---|
| Structure | 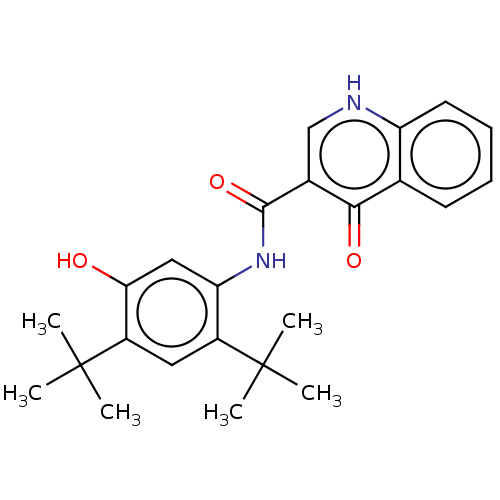
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Froux, L; Elbahnsi, A; Boucherle, B; Billet, A; Baatallah, N; Hoffmann, B; Alliot, J; Zelli, R; Zeinyeh, W; Haudecoeur, R; Chevalier, B; Fortun�, A; Mirval, S; Simard, C; Lehn, P; Mornon, JP; Hinzpeter, A; Becq, F; Callebaut, I; D�cout, JL Targeting different binding sites in the CFTR structures allows to synergistically potentiate channel activity. Eur J Med Chem190:0 (2020) [PubMed] Article
Froux, L; Elbahnsi, A; Boucherle, B; Billet, A; Baatallah, N; Hoffmann, B; Alliot, J; Zelli, R; Zeinyeh, W; Haudecoeur, R; Chevalier, B; Fortun�, A; Mirval, S; Simard, C; Lehn, P; Mornon, JP; Hinzpeter, A; Becq, F; Callebaut, I; D�cout, JL Targeting different binding sites in the CFTR structures allows to synergistically potentiate channel activity. Eur J Med Chem190:0 (2020) [PubMed] Article