| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-adrenergic receptor kinase 2 |
|---|
| Ligand | BDBM50566947 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2102045 (CHEMBL4810441) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Uehling, DE; Joseph, B; Chung, KC; Zhang, AX; Ler, S; Prakesch, MA; Poda, G; Grouleff, J; Aman, A; Kiyota, T; Leung-Hagesteijn, C; Konda, JD; Marcellus, R; Griffin, C; Subramaniam, R; Abibi, A; Strathdee, CA; Isaac, MB; Al-Awar, R; Tiedemann, RE Design, Synthesis, and Characterization of 4-Aminoquinazolines as Potent Inhibitors of the G Protein-Coupled Receptor Kinase 6 (GRK6) for the Treatment of Multiple Myeloma. J Med Chem64:11129-11147 (2021) [PubMed] Article Uehling, DE; Joseph, B; Chung, KC; Zhang, AX; Ler, S; Prakesch, MA; Poda, G; Grouleff, J; Aman, A; Kiyota, T; Leung-Hagesteijn, C; Konda, JD; Marcellus, R; Griffin, C; Subramaniam, R; Abibi, A; Strathdee, CA; Isaac, MB; Al-Awar, R; Tiedemann, RE Design, Synthesis, and Characterization of 4-Aminoquinazolines as Potent Inhibitors of the G Protein-Coupled Receptor Kinase 6 (GRK6) for the Treatment of Multiple Myeloma. J Med Chem64:11129-11147 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-adrenergic receptor kinase 2 |
|---|
| Name: | Beta-adrenergic receptor kinase 2 |
|---|
| Synonyms: | ADRBK2 | ARBK2_HUMAN | BARK2 | GRK3 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 79721.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_615172 |
|---|
| Residue: | 688 |
|---|
| Sequence: | MADLEAVLADVSYLMAMEKSKATPAARASKRIVLPEPSIRSVMQKYLAERNEITFDKIFN
QKIGFLLFKDFCLNEINEAVPQVKFYEEIKEYEKLDNEEDRLCRSRQIYDAYIMKELLSC
SHPFSKQAVEHVQSHLSKKQVTSTLFQPYIEEICESLRGDIFQKFMESDKFTRFCQWKNV
ELNIHLTMNEFSVHRIIGRGGFGEVYGCRKADTGKMYAMKCLDKKRIKMKQGETLALNER
IMLSLVSTGDCPFIVCMTYAFHTPDKLCFILDLMNGGDLHYHLSQHGVFSEKEMRFYATE
IILGLEHMHNRFVVYRDLKPANILLDEHGHARISDLGLACDFSKKKPHASVGTHGYMAPE
VLQKGTAYDSSADWFSLGCMLFKLLRGHSPFRQHKTKDKHEIDRMTLTVNVELPDTFSPE
LKSLLEGLLQRDVSKRLGCHGGGSQEVKEHSFFKGVDWQHVYLQKYPPPLIPPRGEVNAA
DAFDIGSFDEEDTKGIKLLDCDQELYKNFPLVISERWQQEVTETVYEAVNADTDKIEARK
RAKNKQLGHEEDYALGKDCIMHGYMLKLGNPFLTQWQRRYFYLFPNRLEWRGEGESRQNL
LTMEQILSVEETQIKDKKCILFRIKGGKQFVLQCESDPEFVQWKKELNETFKEAQRLLRR
APKFLNKPRSGTVELPKPSLCHRNSNGL
|
|
|
|---|
| BDBM50566947 |
|---|
| n/a |
|---|
| Name | BDBM50566947 |
|---|
| Synonyms: | CHEMBL4847703 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H20FN7O |
|---|
| Mol. Mass. | 393.4175 |
|---|
| SMILES | CCc1cc(Nc2nc(NCc3ccc(F)cn3)nc3cccc(OC)c23)n[nH]1 |
|---|
| Structure | 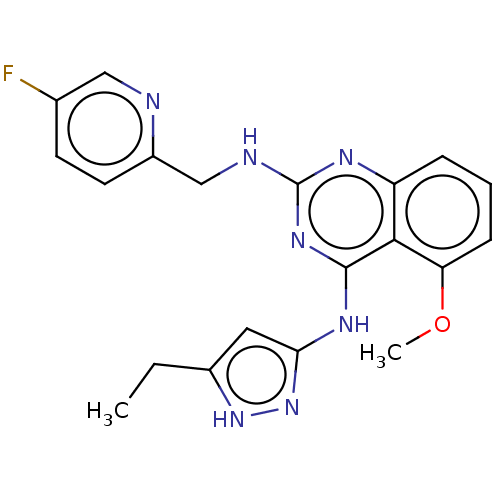
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Uehling, DE; Joseph, B; Chung, KC; Zhang, AX; Ler, S; Prakesch, MA; Poda, G; Grouleff, J; Aman, A; Kiyota, T; Leung-Hagesteijn, C; Konda, JD; Marcellus, R; Griffin, C; Subramaniam, R; Abibi, A; Strathdee, CA; Isaac, MB; Al-Awar, R; Tiedemann, RE Design, Synthesis, and Characterization of 4-Aminoquinazolines as Potent Inhibitors of the G Protein-Coupled Receptor Kinase 6 (GRK6) for the Treatment of Multiple Myeloma. J Med Chem64:11129-11147 (2021) [PubMed] Article
Uehling, DE; Joseph, B; Chung, KC; Zhang, AX; Ler, S; Prakesch, MA; Poda, G; Grouleff, J; Aman, A; Kiyota, T; Leung-Hagesteijn, C; Konda, JD; Marcellus, R; Griffin, C; Subramaniam, R; Abibi, A; Strathdee, CA; Isaac, MB; Al-Awar, R; Tiedemann, RE Design, Synthesis, and Characterization of 4-Aminoquinazolines as Potent Inhibitors of the G Protein-Coupled Receptor Kinase 6 (GRK6) for the Treatment of Multiple Myeloma. J Med Chem64:11129-11147 (2021) [PubMed] Article