| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Activin receptor type-1 |
|---|
| Ligand | BDBM4077 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2107053 (CHEMBL4815728) |
|---|
| IC50 | 4.0±n/a nM |
|---|
| Citation |  Nikhar, S; Siokas, I; Schlicher, L; Lee, S; Gyrd-Hansen, M; Degterev, A; Cuny, GD Design of pyrido[2,3-d]pyrimidin-7-one inhibitors of receptor interacting protein kinase-2 (RIPK2) and nucleotide-binding oligomerization domain (NOD) cell signaling. Eur J Med Chem215:0 (2021) [PubMed] Article Nikhar, S; Siokas, I; Schlicher, L; Lee, S; Gyrd-Hansen, M; Degterev, A; Cuny, GD Design of pyrido[2,3-d]pyrimidin-7-one inhibitors of receptor interacting protein kinase-2 (RIPK2) and nucleotide-binding oligomerization domain (NOD) cell signaling. Eur J Med Chem215:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Activin receptor type-1 |
|---|
| Name: | Activin receptor type-1 |
|---|
| Synonyms: | 2.7.11.30 | ACTR-I | ACVR1 | ACVR1_HUMAN | ACVRLK2 | ALK-2 | ALK2/ACVR1 | Activin receptor type I | Activin receptor-like kinase 2 | Activin receptor-like kinase 2 (ALK-2) | Activin receptor-like kinase 2 (ALK2/ACVR1) | Q04771 | SKR1 | Serine/threonine-protein kinase receptor R1 | TGF-B superfamily receptor type I | TSR-I |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 57158.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 509 |
|---|
| Sequence: | MVDGVMILPVLIMIALPSPSMEDEKPKVNPKLYMCVCEGLSCGNEDHCEGQQCFSSLSIN
DGFHVYQKGCFQVYEQGKMTCKTPPSPGQAVECCQGDWCNRNITAQLPTKGKSFPGTQNF
HLEVGLIILSVVFAVCLLACLLGVALRKFKRRNQERLNPRDVEYGTIEGLITTNVGDSTL
ADLLDHSCTSGSGSGLPFLVQRTVARQITLLECVGKGRYGEVWRGSWQGENVAVKIFSSR
DEKSWFRETELYNTVMLRHENILGFIASDMTSRHSSTQLWLITHYHEMGSLYDYLQLTTL
DTVSCLRIVLSIASGLAHLHIEIFGTQGKPAIAHRDLKSKNILVKKNGQCCIADLGLAVM
HSQSTNQLDVGNNPRVGTKRYMAPEVLDETIQVDCFDSYKRVDIWAFGLVLWEVARRMVS
NGIVEDYKPPFYDVVPNDPSFEDMRKVVCVDQQRPNIPNRWFSDPTLTSLAKLMKECWYQ
NPSARLTALRIKKTLTKIDNSLDKLKTDC
|
|
|
|---|
| BDBM4077 |
|---|
| n/a |
|---|
| Name | BDBM4077 |
|---|
| Synonyms: | 2-({4-[2-(diethylamino)ethoxy]phenyl}amino)-8-ethyl-6-phenyl-7H,8H-pyrido[2,3-d]pyrimidin-7-one | 2-[4-(2-Diethylaminoethoxy)phenylamino]-8-ethyl-6-phenyl-8H-pyrido[2,3-d]pyrimidin-7-one | pyrido[2,3-d]pyrimidin-7-one deriv. 54 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31N5O2 |
|---|
| Mol. Mass. | 457.5673 |
|---|
| SMILES | CCN(CC)CCOc1ccc(Nc2ncc3cc(-c4ccccc4)c(=O)n(CC)c3n2)cc1 |
|---|
| Structure | 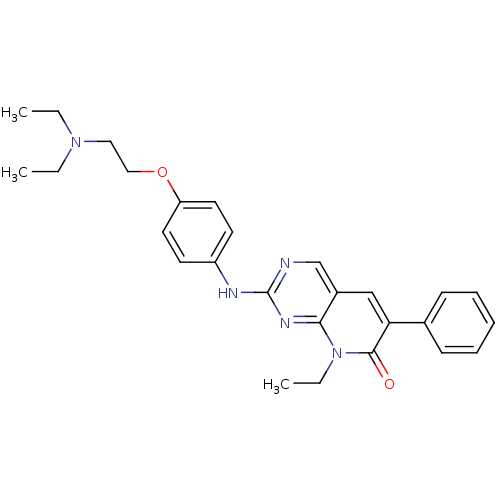
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nikhar, S; Siokas, I; Schlicher, L; Lee, S; Gyrd-Hansen, M; Degterev, A; Cuny, GD Design of pyrido[2,3-d]pyrimidin-7-one inhibitors of receptor interacting protein kinase-2 (RIPK2) and nucleotide-binding oligomerization domain (NOD) cell signaling. Eur J Med Chem215:0 (2021) [PubMed] Article
Nikhar, S; Siokas, I; Schlicher, L; Lee, S; Gyrd-Hansen, M; Degterev, A; Cuny, GD Design of pyrido[2,3-d]pyrimidin-7-one inhibitors of receptor interacting protein kinase-2 (RIPK2) and nucleotide-binding oligomerization domain (NOD) cell signaling. Eur J Med Chem215:0 (2021) [PubMed] Article