| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50133957 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_29405 (CHEMBL643378) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Borthwick, AD; Davies, DE; Ertl, PF; Exall, AM; Haley, TM; Hart, GJ; Jackson, DL; Parry, NR; Patikis, A; Trivedi, N; Weingarten, GG; Woolven, JM Design and synthesis of pyrrolidine-5,5'-trans-lactams (5-oxo-hexahydropyrrolo[3,2-b]pyrroles) as novel mechanism-based inhibitors of human cytomegalovirus protease. 4. Antiviral activity and plasma stability. J Med Chem46:4428-49 (2003) [PubMed] Article Borthwick, AD; Davies, DE; Ertl, PF; Exall, AM; Haley, TM; Hart, GJ; Jackson, DL; Parry, NR; Patikis, A; Trivedi, N; Weingarten, GG; Woolven, JM Design and synthesis of pyrrolidine-5,5'-trans-lactams (5-oxo-hexahydropyrrolo[3,2-b]pyrroles) as novel mechanism-based inhibitors of human cytomegalovirus protease. 4. Antiviral activity and plasma stability. J Med Chem46:4428-49 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_HUMAN | ACHE | Acetylcholinesterase (AChE) | Acetylcholinesterase (human AChE) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 67792.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22303 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPQCLLHTPSLASPLLLLLLWLLGGGVGAEGREDAELLVTVRGGRLRGIRLKTPGGPV
SAFLGIPFAEPPMGPRRFLPPEPKQPWSGVVDATTFQSVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLVWIYGGGFYSGASSLDVYDGRFLVQAERTVLVSM
NYRVGAFGFLALPGSREAPGNVGLLDQRLALQWVQENVAAFGGDPTSVTLFGESAGAASV
GMHLLSPPSRGLFHRAVLQSGAPNGPWATVGMGEARRRATQLAHLVGCPPGGTGGNDTEL
VACLRTRPAQVLVNHEWHVLPQESVFRFSFVPVVDGDFLSDTPEALINAGDFHGLQVLVG
VVKDEGSYFLVYGAPGFSKDNESLISRAEFLAGVRVGVPQVSDLAAEAVVLHYTDWLHPE
DPARLREALSDVVGDHNVVCPVAQLAGRLAAQGARVYAYVFEHRASTLSWPLWMGVPHGY
EIEFIFGIPLDPSRNYTAEEKIFAQRLMRYWANFARTGDPNEPRDPKAPQWPPYTAGAQQ
YVSLDLRPLEVRRGLRAQACAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQDRCSDL
|
|
|
|---|
| BDBM50133957 |
|---|
| n/a |
|---|
| Name | BDBM50133957 |
|---|
| Synonyms: | (S)-2-[(3aS,6S,6aR)-6-Methyl-4-((2R,3S)-2-(S)-methyl-3-methyl-cyclopropanecarbonyl)-5-oxo-hexahydro-pyrrolo[3,2-b]pyrrole-1-carbonyl]-pyrrolidine-1-carboxylic acid (4-isopropyl-phenyl)-amide | CHEMBL139571 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H38N4O4 |
|---|
| Mol. Mass. | 494.6257 |
|---|
| SMILES | CC(C)c1ccc(NC(=O)N2CCC[C@H]2C(=O)N2CC[C@H]3[C@H]2[C@H](C)C(=O)N3C(=O)[C@H]2[C@@H](C)[C@H]2C)cc1 |
|---|
| Structure | 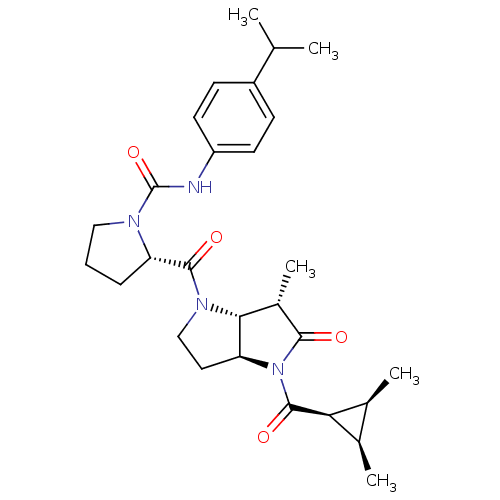
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Borthwick, AD; Davies, DE; Ertl, PF; Exall, AM; Haley, TM; Hart, GJ; Jackson, DL; Parry, NR; Patikis, A; Trivedi, N; Weingarten, GG; Woolven, JM Design and synthesis of pyrrolidine-5,5'-trans-lactams (5-oxo-hexahydropyrrolo[3,2-b]pyrroles) as novel mechanism-based inhibitors of human cytomegalovirus protease. 4. Antiviral activity and plasma stability. J Med Chem46:4428-49 (2003) [PubMed] Article
Borthwick, AD; Davies, DE; Ertl, PF; Exall, AM; Haley, TM; Hart, GJ; Jackson, DL; Parry, NR; Patikis, A; Trivedi, N; Weingarten, GG; Woolven, JM Design and synthesis of pyrrolidine-5,5'-trans-lactams (5-oxo-hexahydropyrrolo[3,2-b]pyrroles) as novel mechanism-based inhibitors of human cytomegalovirus protease. 4. Antiviral activity and plasma stability. J Med Chem46:4428-49 (2003) [PubMed] Article