| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50022775 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2114280 (CHEMBL4823130) |
|---|
| IC50 | 650±n/a nM |
|---|
| Citation |  Yonekawa, MKA; Penteado, BB; Dal'Ongaro Rodrigues, A; Louren�o, EMG; Barbosa, EG; das Neves, SC; de Oliveira, RJ; Marques, MR; Silva, DB; de Lima, DP; Beatriz, A; Oses, JP; Dos S Jaques, JA; Santos, EDAD l-Hypaphorine and d-hypaphorine: Specific antiacetylcholinesterase activity in rat brain tissue. Bioorg Med Chem Lett47:0 (2021) [PubMed] Article Yonekawa, MKA; Penteado, BB; Dal'Ongaro Rodrigues, A; Louren�o, EMG; Barbosa, EG; das Neves, SC; de Oliveira, RJ; Marques, MR; Silva, DB; de Lima, DP; Beatriz, A; Oses, JP; Dos S Jaques, JA; Santos, EDAD l-Hypaphorine and d-hypaphorine: Specific antiacetylcholinesterase activity in rat brain tissue. Bioorg Med Chem Lett47:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_RAT | Acetylcholinesterase (AChE) | Acetylcholinesterase and butyrylcholinesterase (AChE and BChE) | Acetylcholinesterase precursor | Acetylcholinesterase, AChE | Ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 68193.62 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P37136 |
|---|
| Residue: | 614 |
|---|
| Sequence: | MRPPWYPLHTPSLASPLLFLLLSLLGGGARAEGREDPQLLVRVRGGQLRGIRLKAPGGPV
SAFLGIPFAEPPVGSRRFMPPEPKRPWSGILDATTFQNVCYQYVDTLYPGFEGTEMWNPN
RELSEDCLYLNVWTPYPRPTSPTPVLIWIYGGGFYSGASSLDVYDGRFLAQVEGTVLVSM
NYRVGTFGFLALPGSREAPGNVGLLDQRLALQWVQENIAAFGGDPMSVTLFGESAGAASV
GMHILSLPSRSLFHRAVLQSGTPNGPWATVSAGEARRRATLLARLVGCPPGGAGGNDTEL
ISCLRTRPAQDLVDHEWHVLPQESIFRFSFVPVVDGDFLSDTPDALINTGDFQDLQVLVG
VVKDEGSYFLVYGVPGFSKDNESLISRAQFLAGVRIGVPQASDLAAEAVVLHYTDWLHPE
DPAHLRDAMSAVVGDHNVVCPVAQLAGRLAAQGARVYAYIFEHRASTLTWPLWMGVPHGY
EIEFIFGLPLDPSLNYTVEERIFAQRLMQYWTNFARTGDPNDPRDSKSPRWPPYTTAAQQ
YVSLNLKPLEVRRGLRAQTCAFWNRFLPKLLSATDTLDEAERQWKAEFHRWSSYMVHWKN
QFDHYSKQERCSDL
|
|
|
|---|
| BDBM50022775 |
|---|
| n/a |
|---|
| Name | BDBM50022775 |
|---|
| Synonyms: | (m-Hydroxyphenyl)trimethylammonium dimethylcarbamate | 3-Trimethylammoniumphenyl N,N-dimethylcarbamate | 3-[(dimethylcarbamoyl)oxy]-N,N,N-trimethylanilinium | CHEMBL211471 | CHEMBL278020 | Eustigmin | Eustigmine | NEOSTIGMINE | Neostigmine (2) | Prostigmine | Vagostigmine | m-Trimethylammoniumphenyldimethylcarbamate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H19N2O2 |
|---|
| Mol. Mass. | 223.2909 |
|---|
| SMILES | CN(C)C(=O)Oc1cccc(c1)[N+](C)(C)C |
|---|
| Structure | 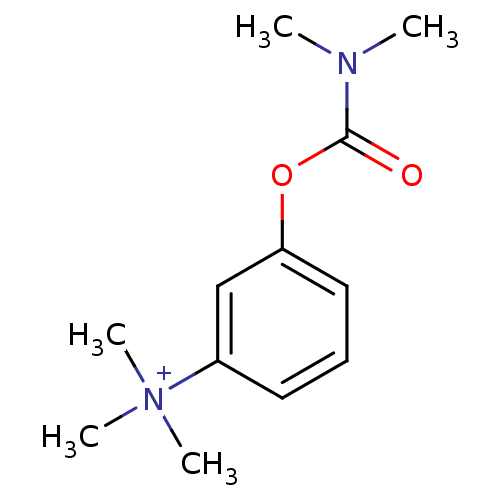
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yonekawa, MKA; Penteado, BB; Dal'Ongaro Rodrigues, A; Louren�o, EMG; Barbosa, EG; das Neves, SC; de Oliveira, RJ; Marques, MR; Silva, DB; de Lima, DP; Beatriz, A; Oses, JP; Dos S Jaques, JA; Santos, EDAD l-Hypaphorine and d-hypaphorine: Specific antiacetylcholinesterase activity in rat brain tissue. Bioorg Med Chem Lett47:0 (2021) [PubMed] Article
Yonekawa, MKA; Penteado, BB; Dal'Ongaro Rodrigues, A; Louren�o, EMG; Barbosa, EG; das Neves, SC; de Oliveira, RJ; Marques, MR; Silva, DB; de Lima, DP; Beatriz, A; Oses, JP; Dos S Jaques, JA; Santos, EDAD l-Hypaphorine and d-hypaphorine: Specific antiacetylcholinesterase activity in rat brain tissue. Bioorg Med Chem Lett47:0 (2021) [PubMed] Article