| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50152233 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303227 (CHEMBL827192) |
|---|
| Ki | 6.5±n/a nM |
|---|
| Citation |  Vu, CB; Shields, P; Peng, B; Kumaravel, G; Jin, X; Phadke, D; Wang, J; Engber, T; Ayyub, E; Petter, RC Triamino derivatives of triazolotriazine and triazolopyrimidine as adenosine A2a receptor antagonists. Bioorg Med Chem Lett14:4835-8 (2004) [PubMed] Article Vu, CB; Shields, P; Peng, B; Kumaravel, G; Jin, X; Phadke, D; Wang, J; Engber, T; Ayyub, E; Petter, RC Triamino derivatives of triazolotriazine and triazolopyrimidine as adenosine A2a receptor antagonists. Bioorg Med Chem Lett14:4835-8 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | AA2AR_RAT | ADENOSINE A2a | Adenosine A2 receptor | Adenosine A2a receptor (A2a) | Adenosine Receptors A2a (A2a) | Adenosine receptor A2a and A3 | Adenosine receptors A2a | Adora2a | Rat striatal adenosine A2a receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 45015.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat A2A receptors expressed in CHO cells. |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGSSVYITVELAIAVLAILGNVLVCWAVWINSNLQNVTNFFVVSLAAADIAVGVLAIPFA
ITISTGFCAACHGCLFFACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGVRAKG
IIAICWVLSFAIGLTPMLGWNNCSQKDGNSTKTCGEGRVTCLFEDVVPMNYMVYYNFFAF
VLLPLLLMLAIYLRIFLAARRQLKQMESQPLPGERTRSTLQKEVHAAKSLAIIVGLFALC
WLPLHIINCFTFFCSTCRHAPPWLMYLAIILSHSNSVVNPFIYAYRIREFRQTFRKIIRT
HVLRRQEPFQAGGSSAWALAAHSTEGEQVSLRLNGHPLGVWANGSATHSGRRPNGYTLGL
GGGGSAQGSPRDVELPTQERQEGQEHPGLRGHLVQARVGASSWSSEFAPS
|
|
|
|---|
| BDBM50152233 |
|---|
| n/a |
|---|
| Name | BDBM50152233 |
|---|
| Synonyms: | CHEMBL363660 | N*7*-{2-[4-(2,4-Difluoro-phenyl)-piperazin-1-yl]-ethyl}-2-furan-2-yl-N*7*-methyl-[1,2,4]triazolo[1,5-c]pyrimidine-5,7-diamine | N7-(2-(4-(2,4-difluorophenyl)piperazin-1-yl)ethyl)-2-(furan-2-yl)-N7-methyl-[1,2,4]triazolo[1,5-c]pyrimidine-5,7-diamine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24F2N8O |
|---|
| Mol. Mass. | 454.4758 |
|---|
| SMILES | CN(CCN1CCN(CC1)c1ccc(F)cc1F)c1cc2nc(nn2c(N)n1)-c1ccco1 |
|---|
| Structure | 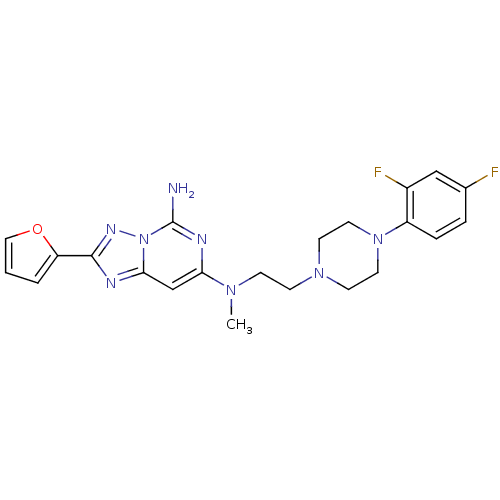
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Vu, CB; Shields, P; Peng, B; Kumaravel, G; Jin, X; Phadke, D; Wang, J; Engber, T; Ayyub, E; Petter, RC Triamino derivatives of triazolotriazine and triazolopyrimidine as adenosine A2a receptor antagonists. Bioorg Med Chem Lett14:4835-8 (2004) [PubMed] Article
Vu, CB; Shields, P; Peng, B; Kumaravel, G; Jin, X; Phadke, D; Wang, J; Engber, T; Ayyub, E; Petter, RC Triamino derivatives of triazolotriazine and triazolopyrimidine as adenosine A2a receptor antagonists. Bioorg Med Chem Lett14:4835-8 (2004) [PubMed] Article