| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A1 |
|---|
| Ligand | BDBM50585333 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2162106 (CHEMBL5046967) |
|---|
| Ki | 1700±n/a nM |
|---|
| Citation |  Bouzriba, C; Chavez Alvarez, AC; Gagn�-Boulet, M; Ouellette, V; Lacroix, J; C�t�, MF; C-Gaudreault, R; Fortin, S Branched alkyl of phenyl 4-(2-oxo-3-alkylimidazolidin-1-yl)benzenesulfonates as unique cytochrome P450 1A1-activated antimitotic prodrugs: Biological evaluation and mechanism of bioactivation. Eur J Med Chem229:0 (2022) [PubMed] Article Bouzriba, C; Chavez Alvarez, AC; Gagn�-Boulet, M; Ouellette, V; Lacroix, J; C�t�, MF; C-Gaudreault, R; Fortin, S Branched alkyl of phenyl 4-(2-oxo-3-alkylimidazolidin-1-yl)benzenesulfonates as unique cytochrome P450 1A1-activated antimitotic prodrugs: Biological evaluation and mechanism of bioactivation. Eur J Med Chem229:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A1 |
|---|
| Name: | Cytochrome P450 1A1 |
|---|
| Synonyms: | CP1A1_HUMAN | CYP1A1 | CYPIA1 | Cytochrome P450 1A | Cytochrome P450 1A1 | Cytochrome P450 1A1 (CYP1A1) | P450 form 6 | P450-C | P450-P1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58177.23 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04798 |
|---|
| Residue: | 512 |
|---|
| Sequence: | MLFPISMSATEFLLASVIFCLVFWVIRASRPQVPKGLKNPPGPWGWPLIGHMLTLGKNPH
LALSRMSQQYGDVLQIRIGSTPVVVLSGLDTIRQALVRQGDDFKGRPDLYTFTLISNGQS
MSFSPDSGPVWAARRRLAQNGLKSFSIASDPASSTSCYLEEHVSKEAEVLISTLQELMAG
PGHFNPYRYVVVSVTNVICAICFGRRYDHNHQELLSLVNLNNNFGEVVGSGNPADFIPIL
RYLPNPSLNAFKDLNEKFYSFMQKMVKEHYKTFEKGHIRDITDSLIEHCQEKQLDENANV
QLSDEKIINIVLDLFGAGFDTVTTAISWSLMYLVMNPRVQRKIQEELDTVIGRSRRPRLS
DRSHLPYMEAFILETFRHSSFVPFTIPHSTTRDTSLKGFYIPKGRCVFVNQWQINHDQKL
WVNPSEFLPERFLTPDGAIDKVLSEKVIIFGMGKRKCIGETIARWEVFLFLAILLQRVEF
SVPLGVKVDMTPIYGLTMKHACCEHFQMQLRS
|
|
|
|---|
| BDBM50585333 |
|---|
| n/a |
|---|
| Name | BDBM50585333 |
|---|
| Synonyms: | CHEMBL5084275 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H28N2O7S |
|---|
| Mol. Mass. | 464.532 |
|---|
| SMILES | COc1cc(OS(=O)(=O)c2ccc(cc2)N2CCN(CC(C)C)C2=O)cc(OC)c1OC |
|---|
| Structure | 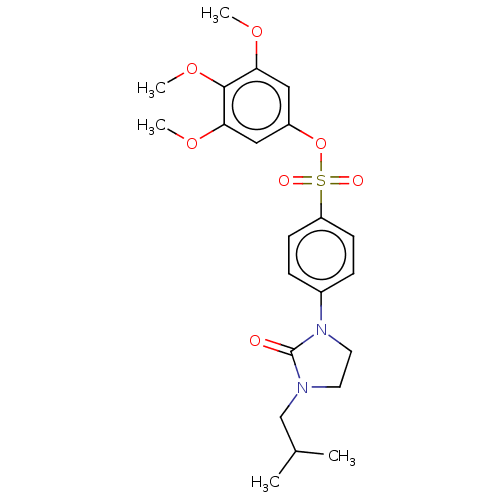
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bouzriba, C; Chavez Alvarez, AC; Gagn�-Boulet, M; Ouellette, V; Lacroix, J; C�t�, MF; C-Gaudreault, R; Fortin, S Branched alkyl of phenyl 4-(2-oxo-3-alkylimidazolidin-1-yl)benzenesulfonates as unique cytochrome P450 1A1-activated antimitotic prodrugs: Biological evaluation and mechanism of bioactivation. Eur J Med Chem229:0 (2022) [PubMed] Article
Bouzriba, C; Chavez Alvarez, AC; Gagn�-Boulet, M; Ouellette, V; Lacroix, J; C�t�, MF; C-Gaudreault, R; Fortin, S Branched alkyl of phenyl 4-(2-oxo-3-alkylimidazolidin-1-yl)benzenesulfonates as unique cytochrome P450 1A1-activated antimitotic prodrugs: Biological evaluation and mechanism of bioactivation. Eur J Med Chem229:0 (2022) [PubMed] Article