| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM200386 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2164346 (CHEMBL5049207) |
|---|
| IC50 | 11000±n/a nM |
|---|
| Citation |  Poddutoori, R; Aardalen, K; Aithal, K; Barahagar, SS; Belliappa, C; Bock, M; Chelur, S; Gerken, A; Gopinath, S; Gruenenfelder, B; Kiffe, M; Krishnaswami, M; Langowski, J; Madapa, S; Narayanan, K; Pandit, C; Panigrahi, SK; Perrone, M; Potakamuri, RK; Ramachandra, M; Ramanathan, A; Ramos, R; Sager, E; Samajdar, S; Subramanya, HS; Thimmasandra, DS; Venetsanakos, E; M�bitz, H Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action. J Med Chem65:4350-4366 (2022) [PubMed] Article Poddutoori, R; Aardalen, K; Aithal, K; Barahagar, SS; Belliappa, C; Bock, M; Chelur, S; Gerken, A; Gopinath, S; Gruenenfelder, B; Kiffe, M; Krishnaswami, M; Langowski, J; Madapa, S; Narayanan, K; Pandit, C; Panigrahi, SK; Perrone, M; Potakamuri, RK; Ramachandra, M; Ramanathan, A; Ramos, R; Sager, E; Samajdar, S; Subramanya, HS; Thimmasandra, DS; Venetsanakos, E; M�bitz, H Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action. J Med Chem65:4350-4366 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM200386 |
|---|
| n/a |
|---|
| Name | BDBM200386 |
|---|
| Synonyms: | US10011599, Example 14 | US9227969, 14 | US9629836, 14 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H24ClFN6O3 |
|---|
| Mol. Mass. | 546.98 |
|---|
| SMILES | Cc1nc2cnc3cc(F)c(cc3c2n1C1CCN(CC1)C(=O)CO)-c1ccc(Oc2ncccn2)cc1Cl |
|---|
| Structure | 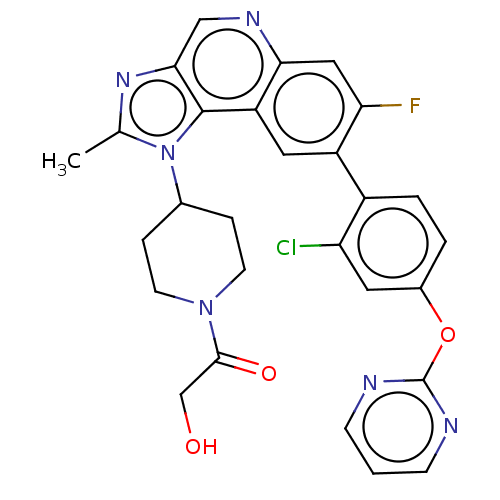
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Poddutoori, R; Aardalen, K; Aithal, K; Barahagar, SS; Belliappa, C; Bock, M; Chelur, S; Gerken, A; Gopinath, S; Gruenenfelder, B; Kiffe, M; Krishnaswami, M; Langowski, J; Madapa, S; Narayanan, K; Pandit, C; Panigrahi, SK; Perrone, M; Potakamuri, RK; Ramachandra, M; Ramanathan, A; Ramos, R; Sager, E; Samajdar, S; Subramanya, HS; Thimmasandra, DS; Venetsanakos, E; M�bitz, H Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action. J Med Chem65:4350-4366 (2022) [PubMed] Article
Poddutoori, R; Aardalen, K; Aithal, K; Barahagar, SS; Belliappa, C; Bock, M; Chelur, S; Gerken, A; Gopinath, S; Gruenenfelder, B; Kiffe, M; Krishnaswami, M; Langowski, J; Madapa, S; Narayanan, K; Pandit, C; Panigrahi, SK; Perrone, M; Potakamuri, RK; Ramachandra, M; Ramanathan, A; Ramos, R; Sager, E; Samajdar, S; Subramanya, HS; Thimmasandra, DS; Venetsanakos, E; M�bitz, H Discovery of MAP855, an Efficacious and Selective MEK1/2 Inhibitor with an ATP-Competitive Mode of Action. J Med Chem65:4350-4366 (2022) [PubMed] Article