| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase |
|---|
| Ligand | BDBM50155839 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302659 (CHEMBL839948) |
|---|
| Ki | 0.03±n/a nM |
|---|
| Citation |  Jarho, EM; Venäläinen, JI; Huuskonen, J; Christiaans, JA; Garcia-Horsman, JA; Forsberg, MM; Järvinen, T; Gynther, J; Männistö, PT; Wallén, EA A cyclopent-2-enecarbonyl group mimics proline at the P2 position of prolyl oligopeptidase inhibitors. J Med Chem47:5605-7 (2004) [PubMed] Article Jarho, EM; Venäläinen, JI; Huuskonen, J; Christiaans, JA; Garcia-Horsman, JA; Forsberg, MM; Järvinen, T; Gynther, J; Männistö, PT; Wallén, EA A cyclopent-2-enecarbonyl group mimics proline at the P2 position of prolyl oligopeptidase inhibitors. J Med Chem47:5605-7 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase |
|---|
| Name: | Prolyl endopeptidase |
|---|
| Synonyms: | 3.4.21.26 | PE | PPCE_PIG | PREP | Post-proline cleaving enzyme |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 80758.04 |
|---|
| Organism: | Sus scrofa |
|---|
| Description: | n/a |
|---|
| Residue: | 710 |
|---|
| Sequence: | MLSFQYPDVYRDETAIQDYHGHKVCDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIR
GLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILS
DDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTH
DGKGMFYNAYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAEL
SDDGRYVLLSIREGCDPVNRLWYCDLQQESNGITGILKWVKLIDNFEGEYDYVTNEGTVF
TFKTNRHSPNYRLINIDFTDPEESKWKVLVPEHEKDVLEWVACVRSNFLVLCYLHDVKNT
LQLHDLATGALLKIFPLEVGSVVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPR
VFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISI
TPNYSVSRLIFVRHMGGVLAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKE
GYTSPKRLTINGGSNGGLLVATCANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYG
CSDSKQHFEWLIKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIV
GRSRKQNNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNIDWIP
|
|
|
|---|
| BDBM50155839 |
|---|
| n/a |
|---|
| Name | BDBM50155839 |
|---|
| Synonyms: | (S)-1-[(R)-2-(4-Phenyl-butyryl)-cyclopent-2-enecarbonyl]-pyrrolidine-2-carbonitrile | CHEMBL185848 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H24N2O2 |
|---|
| Mol. Mass. | 336.4275 |
|---|
| SMILES | O=C(CCCc1ccccc1)C1=CCC[C@H]1C(=O)N1CCC[C@H]1C#N |t:12| |
|---|
| Structure | 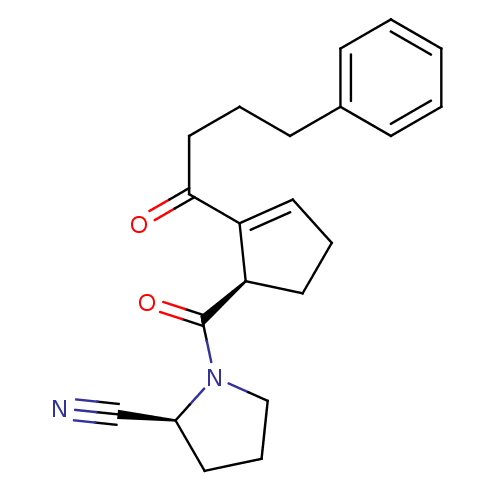
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jarho, EM; Venäläinen, JI; Huuskonen, J; Christiaans, JA; Garcia-Horsman, JA; Forsberg, MM; Järvinen, T; Gynther, J; Männistö, PT; Wallén, EA A cyclopent-2-enecarbonyl group mimics proline at the P2 position of prolyl oligopeptidase inhibitors. J Med Chem47:5605-7 (2004) [PubMed] Article
Jarho, EM; Venäläinen, JI; Huuskonen, J; Christiaans, JA; Garcia-Horsman, JA; Forsberg, MM; Järvinen, T; Gynther, J; Männistö, PT; Wallén, EA A cyclopent-2-enecarbonyl group mimics proline at the P2 position of prolyl oligopeptidase inhibitors. J Med Chem47:5605-7 (2004) [PubMed] Article