| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM50158023 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303336 (CHEMBL840152) |
|---|
| Ki | 1670±n/a nM |
|---|
| Citation |  Cole, DC; Ellingboe, JW; Lennox, WJ; Mazandarani, H; Smith, DL; Stock, JR; Zhang, G; Zhou, P; Schechter, LE N1-arylsulfonyl-3-(1,2,3,6-tetrahydropyridin-4-yl)-1H-indole derivatives are potent and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett15:379-83 (2004) [PubMed] Article Cole, DC; Ellingboe, JW; Lennox, WJ; Mazandarani, H; Smith, DL; Stock, JR; Zhang, G; Zhou, P; Schechter, LE N1-arylsulfonyl-3-(1,2,3,6-tetrahydropyridin-4-yl)-1H-indole derivatives are potent and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett15:379-83 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 46122.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVLSPGQGNNTTSPPAPFETGGNTTGISDVTVSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVKKVEKTGADT
RHGASPAPQPKKSVNGESGSRNWRLGVESKAGGALCANGAVRQGDDGAALEVIEVHRVGN
SKEHLPLPSEAGPTPCAPASFERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPTLLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RQ
|
|
|
|---|
| BDBM50158023 |
|---|
| n/a |
|---|
| Name | BDBM50158023 |
|---|
| Synonyms: | 4-[3-(1,2,3,6-Tetrahydro-pyridin-4-yl)-indole-1-sulfonyl]-phenylamine | CHEMBL444015 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H19N3O2S |
|---|
| Mol. Mass. | 353.438 |
|---|
| SMILES | Nc1ccc(cc1)S(=O)(=O)n1cc(C2=CCNCC2)c2ccccc12 |t:14| |
|---|
| Structure | 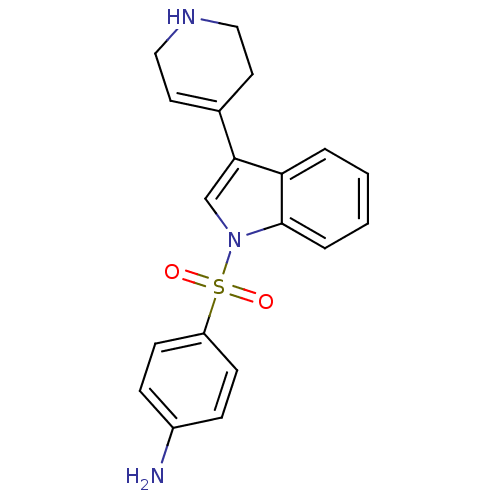
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cole, DC; Ellingboe, JW; Lennox, WJ; Mazandarani, H; Smith, DL; Stock, JR; Zhang, G; Zhou, P; Schechter, LE N1-arylsulfonyl-3-(1,2,3,6-tetrahydropyridin-4-yl)-1H-indole derivatives are potent and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett15:379-83 (2004) [PubMed] Article
Cole, DC; Ellingboe, JW; Lennox, WJ; Mazandarani, H; Smith, DL; Stock, JR; Zhang, G; Zhou, P; Schechter, LE N1-arylsulfonyl-3-(1,2,3,6-tetrahydropyridin-4-yl)-1H-indole derivatives are potent and selective 5-HT6 receptor antagonists. Bioorg Med Chem Lett15:379-83 (2004) [PubMed] Article