| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50137976 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302976 (CHEMBL827955) |
|---|
| Ki | 0.65±n/a nM |
|---|
| Citation |  Campiani, G; Butini, S; Fattorusso, C; Trotta, F; Gemma, S; Catalanotti, B; Nacci, V; Fiorini, I; Cagnotto, A; Mereghetti, I; Mennini, T; Minetti, P; Di Cesare, MA; Stasi, MA; Di Serio, S; Ghirardi, O; Tinti, O; Carminati, P Novel atypical antipsychotic agents: rational design, an efficient palladium-catalyzed route, and pharmacological studies. J Med Chem48:1705-8 (2005) [PubMed] Article Campiani, G; Butini, S; Fattorusso, C; Trotta, F; Gemma, S; Catalanotti, B; Nacci, V; Fiorini, I; Cagnotto, A; Mereghetti, I; Mennini, T; Minetti, P; Di Cesare, MA; Stasi, MA; Di Serio, S; Ghirardi, O; Tinti, O; Carminati, P Novel atypical antipsychotic agents: rational design, an efficient palladium-catalyzed route, and pharmacological studies. J Med Chem48:1705-8 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50137976 |
|---|
| n/a |
|---|
| Name | BDBM50137976 |
|---|
| Synonyms: | 9-(4-Methyl-piperazin-1-yl)-4-thia-10a-aza-benzo[f]azulene | CHEMBL367045 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H19N3S |
|---|
| Mol. Mass. | 297.418 |
|---|
| SMILES | CN1CCN(CC1)C1=Cn2cccc2Sc2ccccc12 |t:8| |
|---|
| Structure | 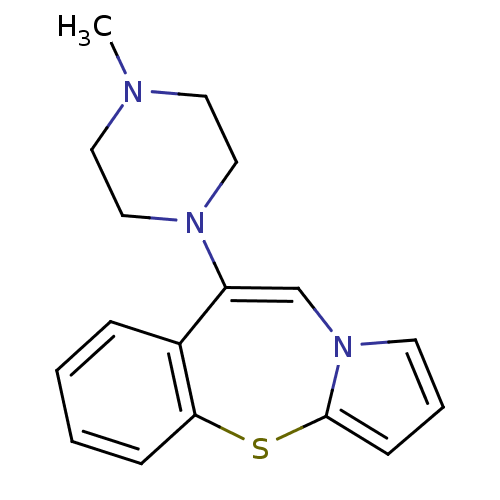
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Campiani, G; Butini, S; Fattorusso, C; Trotta, F; Gemma, S; Catalanotti, B; Nacci, V; Fiorini, I; Cagnotto, A; Mereghetti, I; Mennini, T; Minetti, P; Di Cesare, MA; Stasi, MA; Di Serio, S; Ghirardi, O; Tinti, O; Carminati, P Novel atypical antipsychotic agents: rational design, an efficient palladium-catalyzed route, and pharmacological studies. J Med Chem48:1705-8 (2005) [PubMed] Article
Campiani, G; Butini, S; Fattorusso, C; Trotta, F; Gemma, S; Catalanotti, B; Nacci, V; Fiorini, I; Cagnotto, A; Mereghetti, I; Mennini, T; Minetti, P; Di Cesare, MA; Stasi, MA; Di Serio, S; Ghirardi, O; Tinti, O; Carminati, P Novel atypical antipsychotic agents: rational design, an efficient palladium-catalyzed route, and pharmacological studies. J Med Chem48:1705-8 (2005) [PubMed] Article