| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-2C adrenergic receptor |
|---|
| Ligand | BDBM50163489 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302773 (CHEMBL852482) |
|---|
| Ki | 0.200000±n/a nM |
|---|
| Citation |  Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-2C adrenergic receptor |
|---|
| Name: | Alpha-2C adrenergic receptor |
|---|
| Synonyms: | ADA2C_HUMAN | ADRA2C | ADRA2L2 | ADRA2RL2 | Adrenergic alpha2C | Adrenergic receptor | Adrenergic receptor alpha | Adrenergic, alpha-2C-, receptor | Alpha-2 adrenergic receptor subtype C4 | Alpha-2C adrenoceptor | Alpha-2C adrenoreceptor | adrenergic, alpha-2C-, receptor [Homo sapiens] |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 49552.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P18825 |
|---|
| Residue: | 462 |
|---|
| Sequence: | MASPALAAALAVAAAAGPNASGAGERGSGGVANASGASWGPPRGQYSAGAVAGLAAVVGF
LIVFTVVGNVLVVIAVLTSRALRAPQNLFLVSLASADILVATLVMPFSLANELMAYWYFG
QVWCGVYLALDVLFCTSSIVHLCAISLDRYWSVTQAVEYNLKRTPRRVKATIVAVWLISA
VISFPPLVSLYRQPDGAAYPQCGLNDETWYILSSCIGSFFAPCLIMGLVYARIYRVAKLR
TRTLSEKRAPVGPDGASPTTENGLGAAAGAGENGHCAPPPADVEPDESSAAAERRRRRGA
LRRGGRRRAGAEGGAGGADGQGAGPGAAESGALTASRSPGPGGRLSRASSRSVEFFLSRR
RRARSSVCRRKVAQAREKRFTFVLAVVMGVFVLCWFPFFFSYSLYGICREACQVPGPLFK
FFFWIGYCNSSLNPVIYTVFNQDFRRSFKHILFRRRRRGFRQ
|
|
|
|---|
| BDBM50163489 |
|---|
| n/a |
|---|
| Name | BDBM50163489 |
|---|
| Synonyms: | Acetic acid (3R,3aS)-3-[4-((E)-2-methyl-3-phenyl-allyl)-piperazin-1-ylmethyl]-3a,4-dihydro-3H-chromeno[4,3-c]isoxazol-7-ylester | CHEMBL176116 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H31N3O4 |
|---|
| Mol. Mass. | 461.5527 |
|---|
| SMILES | CC(=O)Oc1ccc2C3=NO[C@@H](CN4CCN(C\C(C)=C\c5ccccc5)CC4)[C@@H]3COc2c1 |t:8| |
|---|
| Structure | 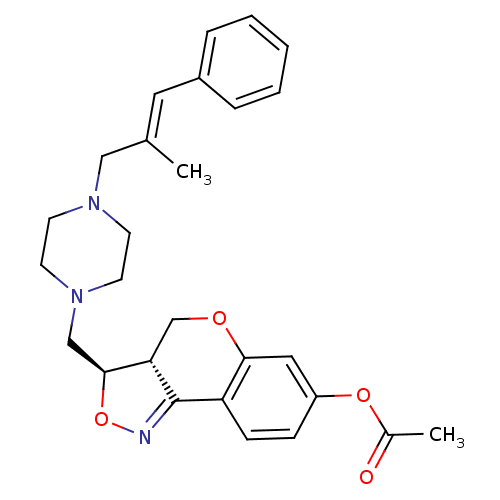
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article
Andrés, JI; Alcázar, J; Alonso, JM; Alvarez, RM; Bakker, MH; Biesmans, I; Cid, JM; De Lucas, AI; Fernández, J; Font, LM; Hens, KA; Iturrino, L; Lenaerts, I; Martínez, S; Megens, AA; Pastor, J; Vermote, PC; Steckler, T Discovery of a new series of centrally active tricyclic isoxazoles combining serotonin (5-HT) reuptake inhibition with alpha2-adrenoceptor blocking activity. J Med Chem48:2054-71 (2005) [PubMed] Article