| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase 2 |
|---|
| Ligand | BDBM11162 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2199775 (CHEMBL5112291) |
|---|
| Ki | >10000±n/a nM |
|---|
| Citation |  Artasensi, A; Angeli, A; Lammi, C; Bollati, C; Gervasoni, S; Baron, G; Matucci, R; Supuran, CT; Vistoli, G; Fumagalli, L Discovery of a Potent and Highly Selective Dipeptidyl Peptidase IV and Carbonic Anhydrase Inhibitor as "Antidiabesity" Agents Based on Repurposing and Morphing of WB-4101. J Med Chem65:13946-13966 (2022) [PubMed] Article Artasensi, A; Angeli, A; Lammi, C; Bollati, C; Gervasoni, S; Baron, G; Matucci, R; Supuran, CT; Vistoli, G; Fumagalli, L Discovery of a Potent and Highly Selective Dipeptidyl Peptidase IV and Carbonic Anhydrase Inhibitor as "Antidiabesity" Agents Based on Repurposing and Morphing of WB-4101. J Med Chem65:13946-13966 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase 2 |
|---|
| Name: | Carbonic anhydrase 2 |
|---|
| Synonyms: | CA-II | CA2 | CAC | CAH2_HUMAN | Carbonate dehydratase II | Carbonic anhydrase 2 (CA II) | Carbonic anhydrase 2 (CA-II) | Carbonic anhydrase 2 (Recombinant CA II) | Carbonic anhydrase C | Carbonic anhydrase II (CA II) | Carbonic anhydrase II (CA-II) | Carbonic anhydrase II (CAII) | Carbonic anhydrase II (hCA II) | Carbonic anhydrase isoenzyme II (hCA II) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 29250.71 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00918 |
|---|
| Residue: | 260 |
|---|
| Sequence: | MSHHWGYGKHNGPEHWHKDFPIAKGERQSPVDIDTHTAKYDPSLKPLSVSYDQATSLRIL
NNGHAFNVEFDDSQDKAVLKGGPLDGTYRLIQFHFHWGSLDGQGSEHTVDKKKYAAELHL
VHWNTKYGDFGKAVQQPDGLAVLGIFLKVGSAKPGLQKVVDVLDSIKTKGKSADFTNFDP
RGLLPESLDYWTYPGSLTTPPLLECVTWIVLKEPISVSSEQVLKFRKLNFNGEGEPEELM
VDNWRPAQPLKNRQIKASFK
|
|
|
|---|
| BDBM11162 |
|---|
| n/a |
|---|
| Name | BDBM11162 |
|---|
| Synonyms: | (1R)-3-oxo-3-[3-(trifluoroethyl)-5,6-dihydro[1,2,4]triazolo[4,3-a]pyrazin-7(8H)-yl]-1-(2,4,5-trifluorobenzyl)propylamine | (3R)-3-amino-1-[3-(trifluoromethyl)-5H,6H,7H,8H-[1,2,4]triazolo[3,4-a]pyrazin-7-yl]-4-(2,4,5-trifluorophenyl)butan-1-one hydrochloride | CHEMBL393336 | MK-0431 | Sitagliptin | Sitagliptin (13) | Triazolopiperazine Analogue 1 | US10479798, Compound MK0431 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H15F6N5O |
|---|
| Mol. Mass. | 407.3136 |
|---|
| SMILES | N[C@@H](CC(=O)N1CCn2c(C1)nnc2C(F)(F)F)Cc1cc(F)c(F)cc1F |r| |
|---|
| Structure | 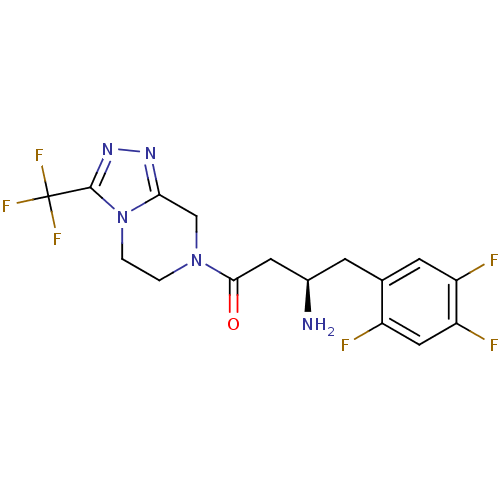
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Artasensi, A; Angeli, A; Lammi, C; Bollati, C; Gervasoni, S; Baron, G; Matucci, R; Supuran, CT; Vistoli, G; Fumagalli, L Discovery of a Potent and Highly Selective Dipeptidyl Peptidase IV and Carbonic Anhydrase Inhibitor as "Antidiabesity" Agents Based on Repurposing and Morphing of WB-4101. J Med Chem65:13946-13966 (2022) [PubMed] Article
Artasensi, A; Angeli, A; Lammi, C; Bollati, C; Gervasoni, S; Baron, G; Matucci, R; Supuran, CT; Vistoli, G; Fumagalli, L Discovery of a Potent and Highly Selective Dipeptidyl Peptidase IV and Carbonic Anhydrase Inhibitor as "Antidiabesity" Agents Based on Repurposing and Morphing of WB-4101. J Med Chem65:13946-13966 (2022) [PubMed] Article