| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Urokinase-type plasminogen activator |
|---|
| Ligand | BDBM50164263 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_302717 (CHEMBL839588) |
|---|
| Ki | >100000±n/a nM |
|---|
| Citation |  Stauffer, KJ; Williams, PD; Selnick, HG; Nantermet, PG; Newton, CL; Homnick, CF; Zrada, MM; Lewis, SD; Lucas, BJ; Krueger, JA; Pietrak, BL; Lyle, EA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Wallace, AA; Sitko, GR; Cook, JJ; Holahan, MA; Stranieri-Michener, M; Leonard, YM; Lynch, JJ; McMasters, DR; Yan, Y 9-hydroxyazafluorenes and their use in thrombin inhibitors. J Med Chem48:2282-93 (2005) [PubMed] Article Stauffer, KJ; Williams, PD; Selnick, HG; Nantermet, PG; Newton, CL; Homnick, CF; Zrada, MM; Lewis, SD; Lucas, BJ; Krueger, JA; Pietrak, BL; Lyle, EA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Wallace, AA; Sitko, GR; Cook, JJ; Holahan, MA; Stranieri-Michener, M; Leonard, YM; Lynch, JJ; McMasters, DR; Yan, Y 9-hydroxyazafluorenes and their use in thrombin inhibitors. J Med Chem48:2282-93 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Urokinase-type plasminogen activator |
|---|
| Name: | Urokinase-type plasminogen activator |
|---|
| Synonyms: | 3.4.21.73 | PLAU | U-plasminogen activator | UROK_HUMAN | Urokinase | Urokinase-type plasminogen activator (uPA) | Urokinase-type plasminogen activator chain B | Urokinase-type plasminogen activator long chain A | Urokinase-type plasminogen activator short chain A | Urokinase-type plasminogen activator/surface receptor | uPA |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48528.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00749 |
|---|
| Residue: | 431 |
|---|
| Sequence: | MRALLARLLLCVLVVSDSKGSNELHQVPSNCDCLNGGTCVSNKYFSNIHWCNCPKKFGGQ
HCEIDKSKTCYEGNGHFYRGKASTDTMGRPCLPWNSATVLQQTYHAHRSDALQLGLGKHN
YCRNPDNRRRPWCYVQVGLKLLVQECMVHDCADGKKPSSPPEELKFQCGQKTLRPRFKII
GGEFTTIENQPWFAAIYRRHRGGSVTYVCGGSLISPCWVISATHCFIDYPKKEDYIVYLG
RSRLNSNTQGEMKFEVENLILHKDYSADTLAHHNDIALLKIRSKEGRCAQPSRTIQTICL
PSMYNDPQFGTSCEITGFGKENSTDYLYPEQLKMTVVKLISHRECQQPHYYGSEVTTKML
CAADPQWKTDSCQGDSGGPLVCSLQGRMTLTGIVSWGRGCALKDKPGVYTRVSHFLPWIR
SHTKEENGLAL
|
|
|
|---|
| BDBM50164263 |
|---|
| n/a |
|---|
| Name | BDBM50164263 |
|---|
| Synonyms: | (S)-1-(9-Hydroxy-9H-3-aza-fluorene-9-carbonyl)-pyrrolidine-2-carboxylic acid 2-aminomethyl-5-chloro-benzylamide | CHEMBL372449 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H25ClN4O3 |
|---|
| Mol. Mass. | 476.955 |
|---|
| SMILES | NCc1ccc(Cl)cc1CNC(=O)[C@@H]1CCCN1C(=O)C1(O)c2ccccc2-c2cnccc12 |
|---|
| Structure | 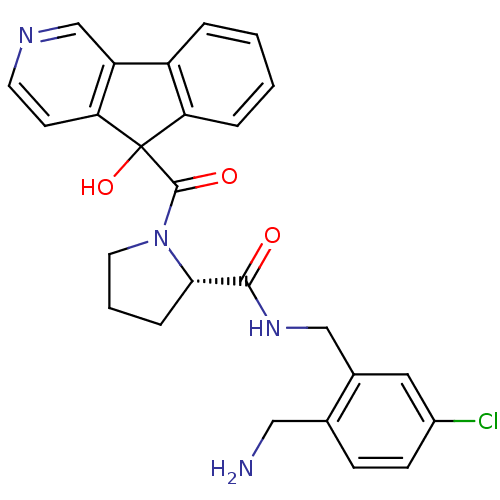
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Stauffer, KJ; Williams, PD; Selnick, HG; Nantermet, PG; Newton, CL; Homnick, CF; Zrada, MM; Lewis, SD; Lucas, BJ; Krueger, JA; Pietrak, BL; Lyle, EA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Wallace, AA; Sitko, GR; Cook, JJ; Holahan, MA; Stranieri-Michener, M; Leonard, YM; Lynch, JJ; McMasters, DR; Yan, Y 9-hydroxyazafluorenes and their use in thrombin inhibitors. J Med Chem48:2282-93 (2005) [PubMed] Article
Stauffer, KJ; Williams, PD; Selnick, HG; Nantermet, PG; Newton, CL; Homnick, CF; Zrada, MM; Lewis, SD; Lucas, BJ; Krueger, JA; Pietrak, BL; Lyle, EA; Singh, R; Miller-Stein, C; White, RB; Wong, B; Wallace, AA; Sitko, GR; Cook, JJ; Holahan, MA; Stranieri-Michener, M; Leonard, YM; Lynch, JJ; McMasters, DR; Yan, Y 9-hydroxyazafluorenes and their use in thrombin inhibitors. J Med Chem48:2282-93 (2005) [PubMed] Article