| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50166631 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_306014 (CHEMBL874551) |
|---|
| IC50 | 1037±n/a nM |
|---|
| Citation |  Le Bourdonnec, B; Ajello, CW; Seida, PR; Susnow, RG; Cassel, JA; Belanger, S; Stabley, GJ; DeHaven, RN; DeHaven-Hudkins, DL; Dolle, RE Arylacetamide kappa opioid receptor agonists with reduced cytochrome P450 2D6 inhibitory activity. Bioorg Med Chem Lett15:2647-52 (2005) [PubMed] Article Le Bourdonnec, B; Ajello, CW; Seida, PR; Susnow, RG; Cassel, JA; Belanger, S; Stabley, GJ; DeHaven, RN; DeHaven-Hudkins, DL; Dolle, RE Arylacetamide kappa opioid receptor agonists with reduced cytochrome P450 2D6 inhibitory activity. Bioorg Med Chem Lett15:2647-52 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50166631 |
|---|
| n/a |
|---|
| Name | BDBM50166631 |
|---|
| Synonyms: | 2-(2-Methanesulfonyl-2,3-dihydro-1H-isoindol-5-yl)-N-methyl-N-((S)-1-phenyl-2-pyrrolidin-1-yl-ethyl)-acetamide | CHEMBL363718 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H31N3O3S |
|---|
| Mol. Mass. | 441.586 |
|---|
| SMILES | CN([C@H](CN1CCCC1)c1ccccc1)C(=O)Cc1ccc2CN(Cc2c1)S(C)(=O)=O |
|---|
| Structure | 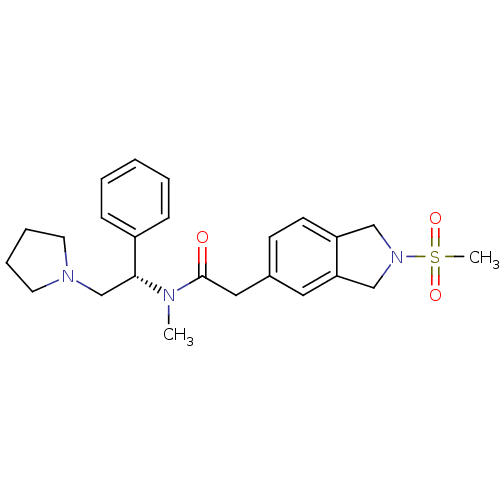
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Le Bourdonnec, B; Ajello, CW; Seida, PR; Susnow, RG; Cassel, JA; Belanger, S; Stabley, GJ; DeHaven, RN; DeHaven-Hudkins, DL; Dolle, RE Arylacetamide kappa opioid receptor agonists with reduced cytochrome P450 2D6 inhibitory activity. Bioorg Med Chem Lett15:2647-52 (2005) [PubMed] Article
Le Bourdonnec, B; Ajello, CW; Seida, PR; Susnow, RG; Cassel, JA; Belanger, S; Stabley, GJ; DeHaven, RN; DeHaven-Hudkins, DL; Dolle, RE Arylacetamide kappa opioid receptor agonists with reduced cytochrome P450 2D6 inhibitory activity. Bioorg Med Chem Lett15:2647-52 (2005) [PubMed] Article