| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Beta-glucosidase A |
|---|
| Ligand | BDBM50166777 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_305320 |
|---|
| IC50 | 52000±n/a nM |
|---|
| Citation |  Łysek, R; Schütz, C; Vogel, P Total asymmetric synthesis of (-)-conduramine B-1 and of its enantiomer. N-Benzyl derivatives of conduramine B-1 are beta-glucosidase inhibitors. Bioorg Med Chem Lett15:3071-5 (2005) [PubMed] Article Łysek, R; Schütz, C; Vogel, P Total asymmetric synthesis of (-)-conduramine B-1 and of its enantiomer. N-Benzyl derivatives of conduramine B-1 are beta-glucosidase inhibitors. Bioorg Med Chem Lett15:3071-5 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Beta-glucosidase A |
|---|
| Name: | Beta-glucosidase A |
|---|
| Synonyms: | BGLS_CALSA | bglA |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 53484.21 |
|---|
| Organism: | Caldocellum saccharolyticum |
|---|
| Description: | ChEMBL_37437 |
|---|
| Residue: | 455 |
|---|
| Sequence: | MDMSFPKGFLWGAATASYQIEGAWNEDGKGESIWDRFTHQKRNILYGHNGDVACDHYHRF
EEDVSLMKELGLKAYRFSIAWTRIFPDGFGTVNQKGLEFYDRLINKLVENGIEPVVTLYH
WDLPQKLQDIGGWANPEIVNYYFDYAMLVINRYKDKVKKWITFNEPYCIAFLGYFHGIHA
PGIKDFKVAMDVVHSLMLSHFKVVKAVKENNIDVEVGITLNLTPVYLQTERLGYKVSEIE
REMVSLSSQLDNQLFLDPVLKGSYPQKLLDYLVQKDLLDSQKALSMQQEVKENFIFPDFL
GINYYTRAVRLYDENSSWIFPIRWEHPAGEYTEMGWEVFPQGLFDLLIWIKESYPQIPIY
ITENGAAYNDIVTEDGKVHDSKRIEYLKQHFEAARKAIENGVDLRGYFVWSLMDNFEWAM
GYTKRFGIIYVDYETQKRIKKDSFYFYQQYIKENS
|
|
|
|---|
| BDBM50166777 |
|---|
| n/a |
|---|
| Name | BDBM50166777 |
|---|
| Synonyms: | 6-(4-Phenoxy-benzylamino)-cyclohex-4-ene-1,2,3-triol | CHEMBL372865 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H21NO4 |
|---|
| Mol. Mass. | 327.3743 |
|---|
| SMILES | OC1C=CC(NCc2ccc(Oc3ccccc3)cc2)C(O)C1O |c:2| |
|---|
| Structure | 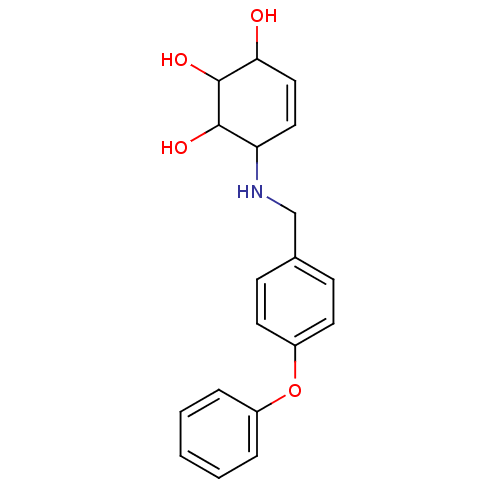
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Łysek, R; Schütz, C; Vogel, P Total asymmetric synthesis of (-)-conduramine B-1 and of its enantiomer. N-Benzyl derivatives of conduramine B-1 are beta-glucosidase inhibitors. Bioorg Med Chem Lett15:3071-5 (2005) [PubMed] Article
Łysek, R; Schütz, C; Vogel, P Total asymmetric synthesis of (-)-conduramine B-1 and of its enantiomer. N-Benzyl derivatives of conduramine B-1 are beta-glucosidase inhibitors. Bioorg Med Chem Lett15:3071-5 (2005) [PubMed] Article