| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Androgen receptor |
|---|
| Ligand | BDBM50099679 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2214673 (CHEMBL5127805) |
|---|
| Ki | 0.300000±n/a nM |
|---|
| Citation |  Xie, Y; Tian, Y; Zhang, Y; Zhang, Z; Chen, R; Li, M; Tang, J; Bian, J; Li, Z; Xu, X Overview of the development of selective androgen receptor modulators (SARMs) as pharmacological treatment for osteoporosis (1998-2021). Eur J Med Chem230:0 (2022) [PubMed] Article Xie, Y; Tian, Y; Zhang, Y; Zhang, Z; Chen, R; Li, M; Tang, J; Bian, J; Li, Z; Xu, X Overview of the development of selective androgen receptor modulators (SARMs) as pharmacological treatment for osteoporosis (1998-2021). Eur J Med Chem230:0 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Androgen receptor |
|---|
| Name: | Androgen receptor |
|---|
| Synonyms: | ANDR_RAT | Androgen receptor (AR) | Ar | Estrogen receptor | Nr3c4 | androgen |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 98219.08 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | P15207 |
|---|
| Residue: | 902 |
|---|
| Sequence: | MEVQLGLGRVYPRPPSKTYRGAFQNLFQSVREAIQNPGPRHPEAASIAPPGACLQQRQET
SPRRRRRQQHPEDGSPQAHIRGTTGYLALEEEQQPSQQQSASEGHPESGCLPEPGAATAP
GKGLPQQPPAPPDQDDSAAPSTLSLLGPTFPGLSSCSADIKDILSEAGTMQLLQQQQQQQ
QQQQQQQQQQQQQQQEVISEGSSSVRAREATGAPSSSKDSYLGGNSTISDSAKELCKAVS
VSMGLGVEALEHLSPGEQLRGDCMYASLLGGPPAVRPTPCAPLAECKGLSLDEGPGKGTE
ETAEYSSFKGGYAKGLEGESLGCSGSSEAGSSGTLEIPSSLSLYKSGAVDEAAAYQNRDY
YNFPLALSGPPHPPPPTHPHARIKLENPSDYGSAWAAAAAQCRYGDLASLHGGSVAGPST
GSPPATASSSWHTLFTAEEGQLYGPGGGGGSSSPSDAGPVAPYGYTRPPQGLASQEGDFS
ASEVWYPGGVVNRVPYPSPSCVKSEMGPWMENYSGPYGDMRLDSTRDHVLPIDYYFPPQK
TCLICGDEASGCHYGALTCGSCKVFFKRAAEGKQKYLCASRNDCTIDKFRRKNCPSCRLR
KCYEAGMTLGARKLKKLGNLKLQEEGENSSAGSPTEDPSQKMTVSHIEGYECQPIFLNVL
EAIEPGVVCAGHDNNQPDSFAALLSSLNELGERQLVHVVKWAKALPGFRNLHVDDQMAVI
QYSWMGLMVFAMGWRSFTNVNSRMLYFAPDLVFNEYRMHKSRMYSQCVRMRHLSQEFGWL
QITPQEFLCMKALLLFSIIPVDGLKNQKFFDELRMNYIKELDRIIACKRKNPTSCSRRFY
QLTKLLDSVQPIARELHQFTFDLLIKSHMVSVDFPEMMAEIISVQVPKILSGKVKPIYFH
TQ
|
|
|
|---|
| BDBM50099679 |
|---|
| n/a |
|---|
| Name | BDBM50099679 |
|---|
| Synonyms: | 3-Bromo-2-hydroxy-2-methyl-N-(4-nitro-3-trifluoromethyl-phenyl)-propionamide | CHEMBL48159 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C11H10BrF3N2O4 |
|---|
| Mol. Mass. | 371.107 |
|---|
| SMILES | C[C@@](O)(CBr)C(=O)Nc1ccc(c(c1)C(F)(F)F)[N+]([O-])=O |
|---|
| Structure | 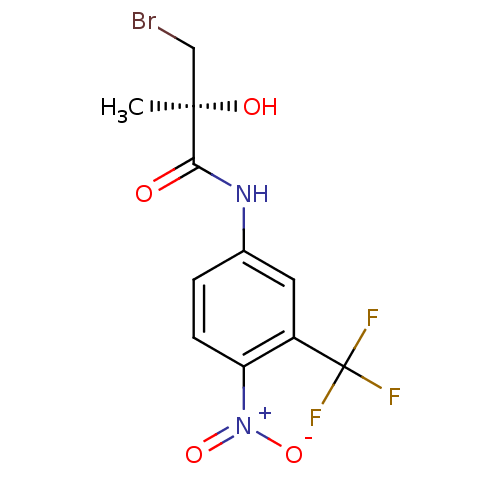
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Xie, Y; Tian, Y; Zhang, Y; Zhang, Z; Chen, R; Li, M; Tang, J; Bian, J; Li, Z; Xu, X Overview of the development of selective androgen receptor modulators (SARMs) as pharmacological treatment for osteoporosis (1998-2021). Eur J Med Chem230:0 (2022) [PubMed] Article
Xie, Y; Tian, Y; Zhang, Y; Zhang, Z; Chen, R; Li, M; Tang, J; Bian, J; Li, Z; Xu, X Overview of the development of selective androgen receptor modulators (SARMs) as pharmacological treatment for osteoporosis (1998-2021). Eur J Med Chem230:0 (2022) [PubMed] Article