| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Transcriptional activator Myb |
|---|
| Ligand | BDBM50595895 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2218219 (CHEMBL5131351) |
|---|
| IC50 | 8.7±n/a nM |
|---|
| Citation |  K�hler, LHF; Reich, S; Yusenko, M; Klempnauer, KH; Shaikh, AH; Ahmed, K; Begemann, G; Schobert, R; Biersack, B A New Naphthopyran Derivative Combines ACS Med Chem Lett13:1783-1790 (2022) [PubMed] Article K�hler, LHF; Reich, S; Yusenko, M; Klempnauer, KH; Shaikh, AH; Ahmed, K; Begemann, G; Schobert, R; Biersack, B A New Naphthopyran Derivative Combines ACS Med Chem Lett13:1783-1790 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Transcriptional activator Myb |
|---|
| Name: | Transcriptional activator Myb |
|---|
| Synonyms: | MYB | MYB_HUMAN | Proto-oncogene c-Myb |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 72345.36 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_120654 |
|---|
| Residue: | 640 |
|---|
| Sequence: | MARRPRHSIYSSDEDDEDFEMCDHDYDGLLPKSGKRHLGKTRWTREEDEKLKKLVEQNGT
DDWKVIANYLPNRTDVQCQHRWQKVLNPELIKGPWTKEEDQRVIELVQKYGPKRWSVIAK
HLKGRIGKQCRERWHNHLNPEVKKTSWTEEEDRIIYQAHKRLGNRWAEIAKLLPGRTDNA
IKNHWNSTMRRKVEQEGYLQESSKASQPAVATSFQKNSHLMGFAQAPPTAQLPATGQPTV
NNDYSYYHISEAQNVSSHVPYPVALHVNIVNVPQPAAAAIQRHYNDEDPEKEKRIKELEL
LLMSTENELKGQQVLPTQNHTCSYPGWHSTTIADHTRPHGDSAPVSCLGEHHSTPSLPAD
PGSLPEESASPARCMIVHQGTILDNVKNLLEFAETLQFIDSFLNTSSNHENSDLEMPSLT
STPLIGHKLTVTTPFHRDQTVKTQKENTVFRTPAIKRSILESSPRTPTPFKHALAAQEIK
YGPLKMLPQTPSHLVEDLQDVIKQESDESGIVAEFQENGPPLLKKIKQEVESPTDKSGNF
FCSHHWEGDSLNTQLFTQTSPVADAPNILTSSVLMAPASEDEDNVLKAFTVPKNRSLASP
LQPCSSTWEPASCGKMEEQMTSSSQARKYVNAFSARTLVM
|
|
|
|---|
| BDBM50595895 |
|---|
| n/a |
|---|
| Name | BDBM50595895 |
|---|
| Synonyms: | CHEMBL321336 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H13N3O3 |
|---|
| Mol. Mass. | 343.3355 |
|---|
| SMILES | NC1=C(C#N)C(c2cccc(c2)[N+]([O-])=O)c2ccc3ccccc3c2O1 |c:1| |
|---|
| Structure | 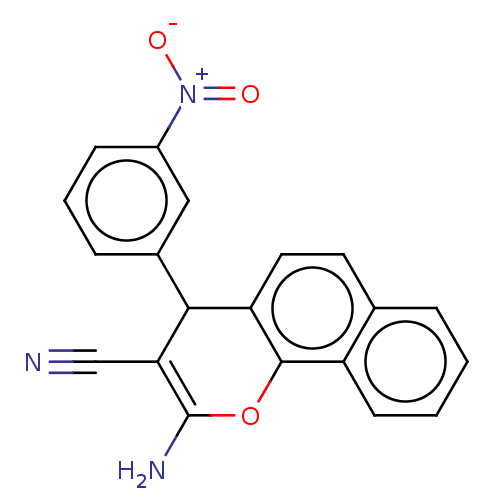
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 K�hler, LHF; Reich, S; Yusenko, M; Klempnauer, KH; Shaikh, AH; Ahmed, K; Begemann, G; Schobert, R; Biersack, B A New Naphthopyran Derivative Combines ACS Med Chem Lett13:1783-1790 (2022) [PubMed] Article
K�hler, LHF; Reich, S; Yusenko, M; Klempnauer, KH; Shaikh, AH; Ahmed, K; Begemann, G; Schobert, R; Biersack, B A New Naphthopyran Derivative Combines ACS Med Chem Lett13:1783-1790 (2022) [PubMed] Article