| Reaction Details |
|---|
 | Report a problem with these data |
| Target | N-formyl peptide receptor 2 |
|---|
| Ligand | BDBM50604088 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2246782 (CHEMBL5160992) |
|---|
| IC50 | 12±n/a nM |
|---|
| Citation |  Mastromarino, M; Favia, M; Schepetkin, IA; Kirpotina, LN; Trojan, E; Niso, M; Carrieri, A; Le?kiewicz, M; Regulska, M; Darida, M; Rossignolo, F; Fontana, S; Quinn, MT; Basta-Kaim, A; Leopoldo, M; Lacivita, E Design, Synthesis, Biological Evaluation, and Computational Studies of Novel Ureidopropanamides as Formyl Peptide Receptor 2 (FPR2) Agonists to Target the Resolution of Inflammation in Central Nervous System Disorders. J Med Chem65:5004-5028 (2022) [PubMed] Article Mastromarino, M; Favia, M; Schepetkin, IA; Kirpotina, LN; Trojan, E; Niso, M; Carrieri, A; Le?kiewicz, M; Regulska, M; Darida, M; Rossignolo, F; Fontana, S; Quinn, MT; Basta-Kaim, A; Leopoldo, M; Lacivita, E Design, Synthesis, Biological Evaluation, and Computational Studies of Novel Ureidopropanamides as Formyl Peptide Receptor 2 (FPR2) Agonists to Target the Resolution of Inflammation in Central Nervous System Disorders. J Med Chem65:5004-5028 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| N-formyl peptide receptor 2 |
|---|
| Name: | N-formyl peptide receptor 2 |
|---|
| Synonyms: | ALXR, FPRL1, FPR2 | FMLP-related receptor I FMLP-R-I | FPR2 | FPR2_HUMAN | FPRH1 | FPRL1 | Formyl Peptide Receptor-Like 1 | HM63 | LXA4 receptor | LXA4R | Lipoxin A4 receptor | Lipoxin A4 receptor (LXA4) | RFP | hFPRL |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 38968.35 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25090 |
|---|
| Residue: | 351 |
|---|
| Sequence: | METNFSTPLNEYEEVSYESAGYTVLRILPLVVLGVTFVLGVLGNGLVIWVAGFRMTRTVT
TICYLNLALADFSFTATLPFLIVSMAMGEKWPFGWFLCKLIHIVVDINLFGSVFLIGFIA
LDRCICVLHPVWAQNHRTVSLAMKVIVGPWILALVLTLPVFLFLTTVTIPNGDTYCTFNF
ASWGGTPEERLKVAITMLTARGIIRFVIGFSLPMSIVAICYGLIAAKIHKKGMIKSSRPL
RVLTAVVASFFICWFPFQLVALLGTVWLKEMLFYGKYKIIDILVNPTSSLAFFNSCLNPM
LYVFVGQDFRERLIHSLPTSLERALSEDSAPTNDTAANSASPPAETELQAM
|
|
|
|---|
| BDBM50604088 |
|---|
| n/a |
|---|
| Name | BDBM50604088 |
|---|
| Synonyms: | CHEMBL5190571 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H23FN4O2 |
|---|
| Mol. Mass. | 442.4848 |
|---|
| SMILES | Fc1ccc(NC(=O)N[C@H](Cc2ccc(cc2)C#N)C(=O)N2CCCc3ccccc23)cc1 |r| |
|---|
| Structure | 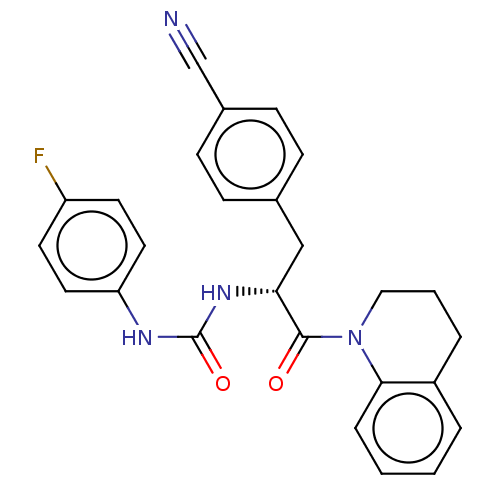
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Mastromarino, M; Favia, M; Schepetkin, IA; Kirpotina, LN; Trojan, E; Niso, M; Carrieri, A; Le?kiewicz, M; Regulska, M; Darida, M; Rossignolo, F; Fontana, S; Quinn, MT; Basta-Kaim, A; Leopoldo, M; Lacivita, E Design, Synthesis, Biological Evaluation, and Computational Studies of Novel Ureidopropanamides as Formyl Peptide Receptor 2 (FPR2) Agonists to Target the Resolution of Inflammation in Central Nervous System Disorders. J Med Chem65:5004-5028 (2022) [PubMed] Article
Mastromarino, M; Favia, M; Schepetkin, IA; Kirpotina, LN; Trojan, E; Niso, M; Carrieri, A; Le?kiewicz, M; Regulska, M; Darida, M; Rossignolo, F; Fontana, S; Quinn, MT; Basta-Kaim, A; Leopoldo, M; Lacivita, E Design, Synthesis, Biological Evaluation, and Computational Studies of Novel Ureidopropanamides as Formyl Peptide Receptor 2 (FPR2) Agonists to Target the Resolution of Inflammation in Central Nervous System Disorders. J Med Chem65:5004-5028 (2022) [PubMed] Article