| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Calpain-1 catalytic subunit |
|---|
| Ligand | BDBM50114340 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_367107 (CHEMBL863671) |
|---|
| IC50 | 22±n/a nM |
|---|
| Citation |  Shirasaki, Y; Nakamura, M; Yamaguchi, M; Miyashita, H; Sakai, O; Inoue, J Exploration of orally available calpain inhibitors 2: peptidyl hemiacetal derivatives. J Med Chem49:3926-32 (2006) [PubMed] Article Shirasaki, Y; Nakamura, M; Yamaguchi, M; Miyashita, H; Sakai, O; Inoue, J Exploration of orally available calpain inhibitors 2: peptidyl hemiacetal derivatives. J Med Chem49:3926-32 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Calpain-1 catalytic subunit |
|---|
| Name: | Calpain-1 catalytic subunit |
|---|
| Synonyms: | CAN1_HUMAN | CANPL1 | CAPN1 | Calpain µ-type | Calpain-1 (u-Calpain) | Calpain-1 catalytic subunit | Calpain1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 81880.51 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 714 |
|---|
| Sequence: | MSEEIITPVYCTGVSAQVQKQRARELGLGRHENAIKYLGQDYEQLRVRCLQSGTLFRDEA
FPPVPQSLGYKDLGPNSSKTYGIKWKRPTELLSNPQFIVDGATRTDICQGALGDCWLLAA
IASLTLNDTLLHRVVPHGQSFQNGYAGIFHFQLWQFGEWVDVVVDDLLPIKDGKLVFVHS
AEGNEFWSALLEKAYAKVNGSYEALSGGSTSEGFEDFTGGVTEWYELRKAPSDLYQIILK
ALERGSLLGCSIDISSVLDMEAITFKKLVKGHAYSVTGAKQVNYRGQVVSLIRMRNPWGE
VEWTGAWSDSSSEWNNVDPYERDQLRVKMEDGEFWMSFRDFMREFTRLEICNLTPDALKS
RTIRKWNTTLYEGTWRRGSTAGGCRNYPATFWVNPQFKIRLDETDDPDDYGDRESGCSFV
LALMQKHRRRERRFGRDMETIGFAVYEVPPELVGQPAVHLKRDFFLANASRARSEQFINL
REVSTRFRLPPGEYVVVPSTFEPNKEGDFVLRFFSEKSAGTVELDDQIQANLPDEQVLSE
EEIDENFKALFRQLAGEDMEISVKELRTILNRIISKHKDLRTKGFSLESCRSMVNLMDRD
GNGKLGLVEFNILWNRIRNYLSIFRKFDLDKSGSMSAYEMRMAIESAGFKLNKKLYELII
TRYSEPDLAVDFDNFVCCLVRLETMFRFFKTLDTDLDGVVTFDLFKWLQLTMFA
|
|
|
|---|
| BDBM50114340 |
|---|
| n/a |
|---|
| Name | BDBM50114340 |
|---|
| Synonyms: | (S)-2-(4-Fluoro-benzenesulfonylamino)-N-((S)-1-formyl-3-methyl-butyl)-3-methyl-butyramide | (S)-2-(4-fluorophenylsulfonamido)-3-methyl-N-((S)-4-methyl-1-oxopentan-2-yl)butanamide | CHEMBL288365 | N-(4-fluorophenylsulfonyl)-L-valyl-L-leucinal | SJA-6017 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H25FN2O4S |
|---|
| Mol. Mass. | 372.455 |
|---|
| SMILES | CC(C)C[C@H](NC(=O)[C@@H](NS(=O)(=O)c1ccc(F)cc1)C(C)C)C=O |r| |
|---|
| Structure | 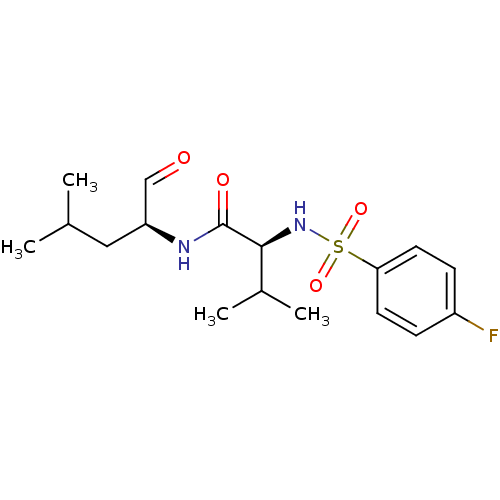
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shirasaki, Y; Nakamura, M; Yamaguchi, M; Miyashita, H; Sakai, O; Inoue, J Exploration of orally available calpain inhibitors 2: peptidyl hemiacetal derivatives. J Med Chem49:3926-32 (2006) [PubMed] Article
Shirasaki, Y; Nakamura, M; Yamaguchi, M; Miyashita, H; Sakai, O; Inoue, J Exploration of orally available calpain inhibitors 2: peptidyl hemiacetal derivatives. J Med Chem49:3926-32 (2006) [PubMed] Article