| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C19 |
|---|
| Ligand | BDBM50197859 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_437625 (CHEMBL905985) |
|---|
| IC50 | 500±n/a nM |
|---|
| Citation |  Garzya, V; Forbes, IT; Gribble, AD; Hadley, MS; Lightfoot, AP; Payne, AH; Smith, AB; Douglas, SE; Cooper, DG; Stansfield, IG; Meeson, M; Dodds, EE; Jones, DN; Wood, M; Reavill, C; Scorer, CA; Worby, A; Riley, G; Eddershaw, P; Ioannou, C; Donati, D; Hagan, JJ; Ratti, EA Studies towards the identification of a new generation of atypical antipsychotic agents. Bioorg Med Chem Lett17:400-5 (2007) [PubMed] Article Garzya, V; Forbes, IT; Gribble, AD; Hadley, MS; Lightfoot, AP; Payne, AH; Smith, AB; Douglas, SE; Cooper, DG; Stansfield, IG; Meeson, M; Dodds, EE; Jones, DN; Wood, M; Reavill, C; Scorer, CA; Worby, A; Riley, G; Eddershaw, P; Ioannou, C; Donati, D; Hagan, JJ; Ratti, EA Studies towards the identification of a new generation of atypical antipsychotic agents. Bioorg Med Chem Lett17:400-5 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C19 |
|---|
| Name: | Cytochrome P450 2C19 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55935.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33261 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKI
YGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFM
ESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYID
LIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFK
KSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50197859 |
|---|
| n/a |
|---|
| Name | BDBM50197859 |
|---|
| Synonyms: | 4-butyl-N-(3-methyl-2,3,4,5-tetrahydro-1H-benzo[d]azepin-7-yl)benzenesulfonamide | CHEMBL231334 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H28N2O2S |
|---|
| Mol. Mass. | 372.524 |
|---|
| SMILES | CCCCc1ccc(cc1)S(=O)(=O)Nc1ccc2CCN(C)CCc2c1 |
|---|
| Structure | 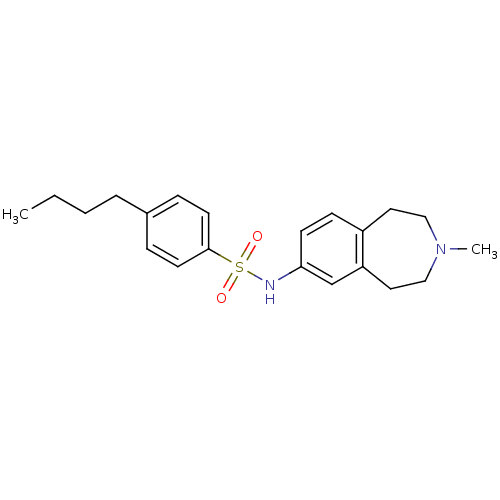
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Garzya, V; Forbes, IT; Gribble, AD; Hadley, MS; Lightfoot, AP; Payne, AH; Smith, AB; Douglas, SE; Cooper, DG; Stansfield, IG; Meeson, M; Dodds, EE; Jones, DN; Wood, M; Reavill, C; Scorer, CA; Worby, A; Riley, G; Eddershaw, P; Ioannou, C; Donati, D; Hagan, JJ; Ratti, EA Studies towards the identification of a new generation of atypical antipsychotic agents. Bioorg Med Chem Lett17:400-5 (2007) [PubMed] Article
Garzya, V; Forbes, IT; Gribble, AD; Hadley, MS; Lightfoot, AP; Payne, AH; Smith, AB; Douglas, SE; Cooper, DG; Stansfield, IG; Meeson, M; Dodds, EE; Jones, DN; Wood, M; Reavill, C; Scorer, CA; Worby, A; Riley, G; Eddershaw, P; Ioannou, C; Donati, D; Hagan, JJ; Ratti, EA Studies towards the identification of a new generation of atypical antipsychotic agents. Bioorg Med Chem Lett17:400-5 (2007) [PubMed] Article