| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glutamate carboxypeptidase 2 |
|---|
| Ligand | BDBM50610873 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2282296 |
|---|
| IC50 | 7.1±n/a nM |
|---|
| Citation |  Petrov, SA; Zyk, NY; Machulkin, AE; Beloglazkina, EK; Majouga, AG PSMA-targeted low-molecular double conjugates for diagnostics and therapy. Eur J Med Chem225:0 (2021) [PubMed] Petrov, SA; Zyk, NY; Machulkin, AE; Beloglazkina, EK; Majouga, AG PSMA-targeted low-molecular double conjugates for diagnostics and therapy. Eur J Med Chem225:0 (2021) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glutamate carboxypeptidase 2 |
|---|
| Name: | Glutamate carboxypeptidase 2 |
|---|
| Synonyms: | FGCP | FOLH | FOLH1 | FOLH1_HUMAN | Folate hydrolase 1 | Folylpoly-gamma-glutamate carboxypeptidase | Glutamate carboxypeptidase 2 | Glutamate carboxypeptidase II | Membrane glutamate carboxypeptidase | N-acetylated-alpha-linked acidic dipeptidase I | NAALAD1 | NAALADase I | PSM | PSMA | Prostate-specific membrane antigen | Pteroylpoly-gamma-glutamate carboxypeptidase | mGCP |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 84333.66 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1497035 |
|---|
| Residue: | 750 |
|---|
| Sequence: | MWNLLHETDSAVATARRPRWLCAGALVLAGGFFLLGFLFGWFIKSSNEATNITPKHNMKA
FLDELKAENIKKFLYNFTQIPHLAGTEQNFQLAKQIQSQWKEFGLDSVELAHYDVLLSYP
NKTHPNYISIINEDGNEIFNTSLFEPPPPGYENVSDIVPPFSAFSPQGMPEGDLVYVNYA
RTEDFFKLERDMKINCSGKIVIARYGKVFRGNKVKNAQLAGAKGVILYSDPADYFAPGVK
SYPDGWNLPGGGVQRGNILNLNGAGDPLTPGYPANEYAYRRGIAEAVGLPSIPVHPIGYY
DAQKLLEKMGGSAPPDSSWRGSLKVPYNVGPGFTGNFSTQKVKMHIHSTNEVTRIYNVIG
TLRGAVEPDRYVILGGHRDSWVFGGIDPQSGAAVVHEIVRSFGTLKKEGWRPRRTILFAS
WDAEEFGLLGSTEWAEENSRLLQERGVAYINADSSIEGNYTLRVDCTPLMYSLVHNLTKE
LKSPDEGFEGKSLYESWTKKSPSPEFSGMPRISKLGSGNDFEVFFQRLGIASGRARYTKN
WETNKFSGYPLYHSVYETYELVEKFYDPMFKYHLTVAQVRGGMVFELANSIVLPFDCRDY
AVVLRKYADKIYSISMKHPQEMKTYSVSFDSLFSAVKNFTEIASKFSERLQDFDKSNPIV
LRMMNDQLMFLERAFIDPLGLPDRPFYRHVIYAPSSHNKYAGESFPGIYDALFDIESKVD
PSKAWGEVKRQIYVAAFTVQAAAETLSEVA
|
|
|
|---|
| BDBM50610873 |
|---|
| n/a |
|---|
| Name | BDBM50610873 |
|---|
| Synonyms: | CHEMBL5277672 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C62H96FN11O26Si |
|---|
| Mol. Mass. | 1458.5676 |
|---|
| SMILES | [#6]C([#6])([#6])[Si;v4](F)(c1ccc(cc1)-[#6](=O)-[#7]-[#6]-[#6@@H](-[#7]-[#6](=O)-[#6]-[#6](-[#7]-1-[#6]-[#6]-[#7](-[#6]-[#6](-[#8])=O)-[#6]-[#6]-[#7](-[#6]-[#6](-[#8])=O)-[#6]-[#6]-[#7](-[#6]-[#6](-[#8])=O)-[#6]-[#6]-1)-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7]-[#6](=O)-[#6]-[#6]-[#6](=O)-[#7]-[#6]-[#6]-[#6]-[#6@@H](-[#7]-[#6](=O)-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#8]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](-[#8])=O)-[#6](-[#8])=O)-[#6](-[#8])=O)-[#6](-[#8])=O)C([#6])([#6])[#6] |r| |
|---|
| Structure | 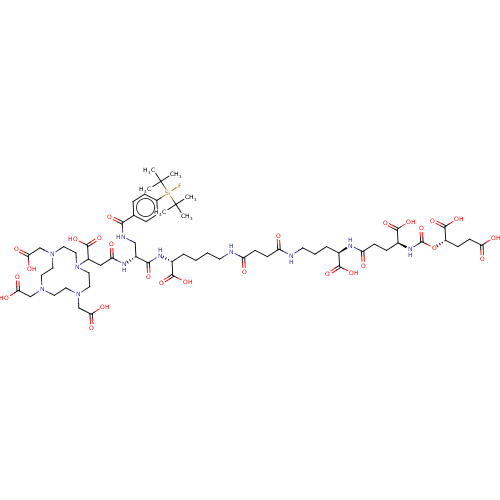
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Petrov, SA; Zyk, NY; Machulkin, AE; Beloglazkina, EK; Majouga, AG PSMA-targeted low-molecular double conjugates for diagnostics and therapy. Eur J Med Chem225:0 (2021) [PubMed]
Petrov, SA; Zyk, NY; Machulkin, AE; Beloglazkina, EK; Majouga, AG PSMA-targeted low-molecular double conjugates for diagnostics and therapy. Eur J Med Chem225:0 (2021) [PubMed]