| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2A6 |
|---|
| Ligand | BDBM50613944 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2294775 |
|---|
| Ki | 50±n/a nM |
|---|
| Citation |  Yamaguchi, Y; Nishizono, N; Kobayashi, D; Yoshimura, T; Wada, K; Kobayashi, K; Oda, K Synthesis and biological evaluation of coumarin derivatives as selective CYP2A6 inhibitors. Bioorg Med Chem Lett86:0 (2023) [PubMed] Yamaguchi, Y; Nishizono, N; Kobayashi, D; Yoshimura, T; Wada, K; Kobayashi, K; Oda, K Synthesis and biological evaluation of coumarin derivatives as selective CYP2A6 inhibitors. Bioorg Med Chem Lett86:0 (2023) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2A6 |
|---|
| Name: | Cytochrome P450 2A6 |
|---|
| Synonyms: | 1,4-cineole 2-exo-monooxygenase | 1.14.13.- | CP2A6_HUMAN | CYP2A3 | CYP2A6 | CYPIIA6 | Coumarin 7-hydroxylase | Cytochrome P450 2A6 | Cytochrome P450 IIA3 | Cytochrome P450(I) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 56514.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11509 |
|---|
| Residue: | 494 |
|---|
| Sequence: | MLASGMLLVALLVCLTVMVLMSVWQQRKSKGKLPPGPTPLPFIGNYLQLNTEQMYNSLMK
ISERYGPVFTIHLGPRRVVVLCGHDAVREALVDQAEEFSGRGEQATFDWVFKGYGVVFSN
GERAKQLRRFSIATLRDFGVGKRGIEERIQEEAGFLIDALRGTGGANIDPTFFLSRTVSN
VISSIVFGDRFDYKDKEFLSLLRMMLGIFQFTSTSTGQLYEMFSSVMKHLPGPQQQAFQL
LQGLEDFIAKKVEHNQRTLDPNSPRDFIDSFLIRMQEEEKNPNTEFYLKNLVMTTLNLFI
GGTETVSTTLRYGFLLLMKHPEVEAKVHEEIDRVIGKNRQPKFEDRAKMPYMEAVIHEIQ
RFGDVIPMSLARRVKKDTKFRDFFLPKGTEVYPMLGSVLRDPSFFSNPQDFNPQHFLNEK
GQFKKSDAFVPFSIGKRNCFGEGLARMELFLFFTTVMQNFRLKSSQSPKDIDVSPKHVGF
ATIPRNYTMSFLPR
|
|
|
|---|
| BDBM50613944 |
|---|
| n/a |
|---|
| Name | BDBM50613944 |
|---|
| Synonyms: | CHEMBL502746 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H8O3 |
|---|
| Mol. Mass. | 176.1687 |
|---|
| SMILES | COc1cccc2oc(=O)ccc12 |
|---|
| Structure | 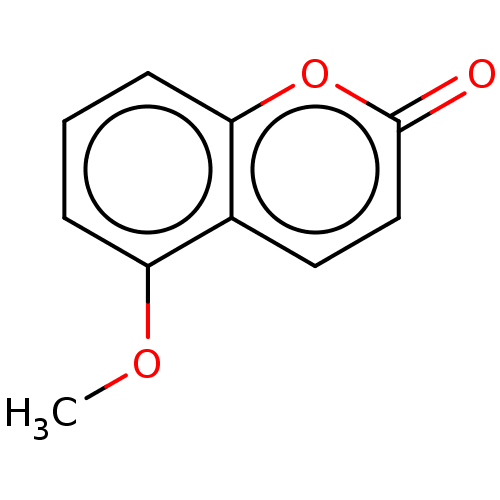
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yamaguchi, Y; Nishizono, N; Kobayashi, D; Yoshimura, T; Wada, K; Kobayashi, K; Oda, K Synthesis and biological evaluation of coumarin derivatives as selective CYP2A6 inhibitors. Bioorg Med Chem Lett86:0 (2023) [PubMed]
Yamaguchi, Y; Nishizono, N; Kobayashi, D; Yoshimura, T; Wada, K; Kobayashi, K; Oda, K Synthesis and biological evaluation of coumarin derivatives as selective CYP2A6 inhibitors. Bioorg Med Chem Lett86:0 (2023) [PubMed]