| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor gamma |
|---|
| Ligand | BDBM50213186 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_447438 (CHEMBL896461) |
|---|
| EC50 | 350±n/a nM |
|---|
| Citation |  Kim, NJ; Lee, KO; Koo, BW; Li, F; Yoo, JK; Park, HJ; Min, KH; Lim, JI; Kim, MK; Kim, JK; Suh, YG Design, synthesis, and structure-activity relationship of carbamate-tethered aryl propanoic acids as novel PPARalpha/gamma dual agonists. Bioorg Med Chem Lett17:3595-8 (2007) [PubMed] Article Kim, NJ; Lee, KO; Koo, BW; Li, F; Yoo, JK; Park, HJ; Min, KH; Lim, JI; Kim, MK; Kim, JK; Suh, YG Design, synthesis, and structure-activity relationship of carbamate-tethered aryl propanoic acids as novel PPARalpha/gamma dual agonists. Bioorg Med Chem Lett17:3595-8 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor gamma |
|---|
| Name: | Peroxisome proliferator-activated receptor gamma |
|---|
| Synonyms: | NR1C3 | Nuclear receptor subfamily 1 group C member 3 | PPAR-gamma | PPARG | PPARG_HUMAN | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor gamma (PPAR gamma) | Peroxisome proliferator-activated receptor gamma (PPARG) | Peroxisome proliferator-activated receptor gamma (PPARγ) | Peroxisome proliferator-activated receptor gamma/Nuclear receptor corepressor 2 | peroxisome proliferator-activated receptor gamma isoform 2 |
|---|
| Type: | Nuclear Receptor |
|---|
| Mol. Mass.: | 57613.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P37231 |
|---|
| Residue: | 505 |
|---|
| Sequence: | MGETLGDSPIDPESDSFTDTLSANISQEMTMVDTEMPFWPTNFGISSVDLSVMEDHSHSF
DIKPFTTVDFSSISTPHYEDIPFTRTDPVVADYKYDLKLQEYQSAIKVEPASPPYYSEKT
QLYNKPHEEPSNSLMAIECRVCGDKASGFHYGVHACEGCKGFFRRTIRLKLIYDRCDLNC
RIHKKSRNKCQYCRFQKCLAVGMSHNAIRFGRMPQAEKEKLLAEISSDIDQLNPESADLR
ALAKHLYDSYIKSFPLTKAKARAILTGKTTDKSPFVIYDMNSLMMGEDKIKFKHITPLQE
QSKEVAIRIFQGCQFRSVEAVQEITEYAKSIPGFVNLDLNDQVTLLKYGVHEIIYTMLAS
LMNKDGVLISEGQGFMTREFLKSLRKPFGDFMEPKFEFAVKFNALELDDSDLAIFIAVII
LSGDRPGLLNVKPIEDIQDNLLQALELQLKLNHPESSQLFAKLLQKMTDLRQIVTEHVQL
LQVIKKTETDMSLHPLLQEIYKDLY
|
|
|
|---|
| BDBM50213186 |
|---|
| n/a |
|---|
| Name | BDBM50213186 |
|---|
| Synonyms: | (2R)-3-(4-(3-(benzylcarbamothioyloxy)-3-phenylpropyl)phenyl)-2-ethoxypropanoic acid | CHEMBL394031 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H31NO4S |
|---|
| Mol. Mass. | 477.615 |
|---|
| SMILES | CCO[C@H](Cc1ccc(CCC(OC(=S)NCc2ccccc2)c2ccccc2)cc1)C(O)=O |
|---|
| Structure | 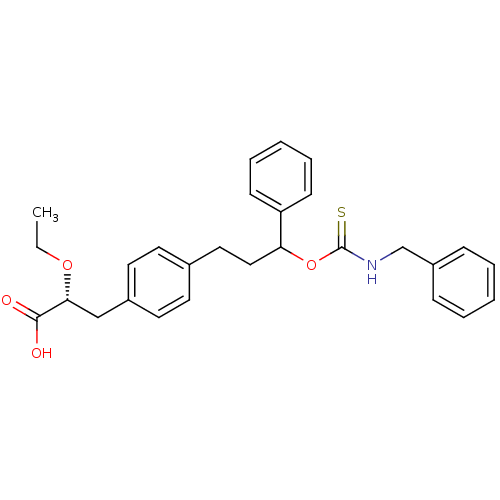
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kim, NJ; Lee, KO; Koo, BW; Li, F; Yoo, JK; Park, HJ; Min, KH; Lim, JI; Kim, MK; Kim, JK; Suh, YG Design, synthesis, and structure-activity relationship of carbamate-tethered aryl propanoic acids as novel PPARalpha/gamma dual agonists. Bioorg Med Chem Lett17:3595-8 (2007) [PubMed] Article
Kim, NJ; Lee, KO; Koo, BW; Li, F; Yoo, JK; Park, HJ; Min, KH; Lim, JI; Kim, MK; Kim, JK; Suh, YG Design, synthesis, and structure-activity relationship of carbamate-tethered aryl propanoic acids as novel PPARalpha/gamma dual agonists. Bioorg Med Chem Lett17:3595-8 (2007) [PubMed] Article