| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fatty-acid amide hydrolase 1 [30-579] |
|---|
| Ligand | BDBM50218952 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_445266 (CHEMBL894412) |
|---|
| IC50 | 47±n/a nM |
|---|
| Citation |  Myllymäki, MJ; Saario, SM; Kataja, AO; Castillo-Melendez, JA; Nevalainen, T; Juvonen, RO; Järvinen, T; Koskinen, AM Design, synthesis, and in vitro evaluation of carbamate derivatives of 2-benzoxazolyl- and 2-benzothiazolyl-(3-hydroxyphenyl)-methanones as novel fatty acid amide hydrolase inhibitors. J Med Chem50:4236-42 (2007) [PubMed] Article Myllymäki, MJ; Saario, SM; Kataja, AO; Castillo-Melendez, JA; Nevalainen, T; Juvonen, RO; Järvinen, T; Koskinen, AM Design, synthesis, and in vitro evaluation of carbamate derivatives of 2-benzoxazolyl- and 2-benzothiazolyl-(3-hydroxyphenyl)-methanones as novel fatty acid amide hydrolase inhibitors. J Med Chem50:4236-42 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Fatty-acid amide hydrolase 1 [30-579] |
|---|
| Name: | Fatty-acid amide hydrolase 1 [30-579] |
|---|
| Synonyms: | Anandamide amidohydrolase 1 | FAAH1_RAT | Faah | Faah1 | Fatty Acid Amide Hydrolase | Fatty Acid Amide Hydrolic, FAAH | Fatty-acid amide hydrolase (FAAH) | Fatty-acid amide hydrolase 1 | Fatty-acid amide hydrolase 1 (FAAH) | Fatty-acid amide hydrolase 1 (aa 30-579) | Oleamide hydrolase 1 |
|---|
| Type: | Single-pass membrane protein; homodimer |
|---|
| Mol. Mass.: | 60474.00 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P97612 (aa 30-579) |
|---|
| Residue: | 550 |
|---|
| Sequence: | RWTGRQKARGAATRARQKQRASLETMDKAVQRFRLQNPDLDSEALLTLPLLQLVQKLQSG
ELSPEAVFFTYLGKAWEVNKGTNCVTSYLTDCETQLSQAPRQGLLYGVPVSLKECFSYKG
HDSTLGLSLNEGMPSESDCVVVQVLKLQGAVPFVHTNVPQSMLSFDCSNPLFGQTMNPWK
SSKSPGGSSGGEGALIGSGGSPLGLGTDIGGSIRFPSAFCGICGLKPTGNRLSKSGLKGC
VYGQTAVQLSLGPMARDVESLALCLKALLCEHLFTLDPTVPPLPFREEVYRSSRPLRVGY
YETDNYTMPSPAMRRALIETKQRLEAAGHTLIPFLPNNIPYALEVLSAGGLFSDGGRSFL
QNFKGDFVDPCLGDLILILRLPSWFKRLLSLLLKPLFPRLAAFLNSMRPRSAEKLWKLQH
EIEMYRQSVIAQWKAMNLDVLLTPMLGPALDLNTPGRATGAISYTVLYNCLDFPAGVVPV
TTVTAEDDAQMELYKGYFGDIWDIILKKAMKNSVGLPVAVQCVALPWQEELCLRFMREVE
QLMTPQKQPS
|
|
|
|---|
| BDBM50218952 |
|---|
| n/a |
|---|
| Name | BDBM50218952 |
|---|
| Synonyms: | 3-(benzo[d]thiazole-2-carbonyl)phenyl cyclopentylcarbamate | CHEMBL389742 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H18N2O3S |
|---|
| Mol. Mass. | 366.434 |
|---|
| SMILES | O=C(NC1CCCC1)Oc1cccc(c1)C(=O)c1nc2ccccc2s1 |
|---|
| Structure | 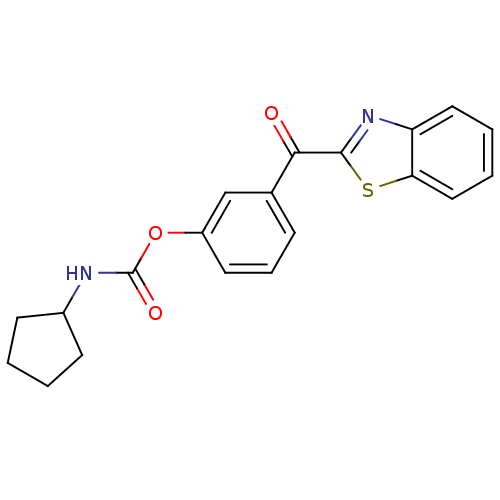
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Myllymäki, MJ; Saario, SM; Kataja, AO; Castillo-Melendez, JA; Nevalainen, T; Juvonen, RO; Järvinen, T; Koskinen, AM Design, synthesis, and in vitro evaluation of carbamate derivatives of 2-benzoxazolyl- and 2-benzothiazolyl-(3-hydroxyphenyl)-methanones as novel fatty acid amide hydrolase inhibitors. J Med Chem50:4236-42 (2007) [PubMed] Article
Myllymäki, MJ; Saario, SM; Kataja, AO; Castillo-Melendez, JA; Nevalainen, T; Juvonen, RO; Järvinen, T; Koskinen, AM Design, synthesis, and in vitro evaluation of carbamate derivatives of 2-benzoxazolyl- and 2-benzothiazolyl-(3-hydroxyphenyl)-methanones as novel fatty acid amide hydrolase inhibitors. J Med Chem50:4236-42 (2007) [PubMed] Article