| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen synthase kinase-3 beta |
|---|
| Ligand | BDBM50220373 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_457307 (CHEMBL941840) |
|---|
| IC50 | 15±n/a nM |
|---|
| Citation |  Shin, D; Lee, SC; Heo, YS; Lee, WY; Cho, YS; Kim, YE; Hyun, YL; Cho, JM; Lee, YS; Ro, S Design and synthesis of 7-hydroxy-1H-benzoimidazole derivatives as novel inhibitors of glycogen synthase kinase-3beta. Bioorg Med Chem Lett17:5686-9 (2007) [PubMed] Article Shin, D; Lee, SC; Heo, YS; Lee, WY; Cho, YS; Kim, YE; Hyun, YL; Cho, JM; Lee, YS; Ro, S Design and synthesis of 7-hydroxy-1H-benzoimidazole derivatives as novel inhibitors of glycogen synthase kinase-3beta. Bioorg Med Chem Lett17:5686-9 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen synthase kinase-3 beta |
|---|
| Name: | Glycogen synthase kinase-3 beta |
|---|
| Synonyms: | GSK-3 beta | GSK-3, beta | GSK3B | GSK3B_HUMAN | Glycogen synthase kinase 3 beta (GSK3B) | Glycogen synthase kinase 3-beta (GSK3B) | Glycogen synthase kinase-3 beta (GSK-3B) | Glycogen synthase kinase-3 beta (GSK3 Beta) | Glycogen synthase kinase-3 beta (GSK3B) | Glycogen synthase kinase-3B (GSK-3B) | Glycogen synthase kinase-3beta (GSK3B) | Serine/threonine-protein kinase GSK3B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 46756.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49841 |
|---|
| Residue: | 420 |
|---|
| Sequence: | MSGRPRTTSFAESCKPVQQPSAFGSMKVSRDKDGSKVTTVVATPGQGPDRPQEVSYTDTK
VIGNGSFGVVYQAKLCDSGELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFYSSG
EKKDEVYLNLVLDYVPETVYRVARHYSRAKQTLPVIYVKLYMYQLFRSLAYIHSFGICHR
DIKPQNLLLDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSSIDV
WSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIKAHP
WTKVFRPRTPPEAIALCSRLLEYTPTARLTPLEACAHSFFDELRDPNVKLPNGRDTPALF
NFTTQELSSNPPLATILIPPHARIQAAASTPTNATAASDANTGDRGQTNNAASASASNST
|
|
|
|---|
| BDBM50220373 |
|---|
| n/a |
|---|
| Name | BDBM50220373 |
|---|
| Synonyms: | 2-(2,4-DICHLORO-PHENYL)-7-HYDROXY-1H-BENZOIMIDAZOLE-4-CARBOXYLIC ACID [2-(4-METHANESULFONYLAMINO-PHENYL)-ETHYL]-AMIDE | CHEMBL436837 | N-(4-(methylsulfonamido)phenethyl)-2-(2,4-dichlorophenyl)-7-hydroxy-1H-benzo[d]imidazole-4-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H20Cl2N4O4S |
|---|
| Mol. Mass. | 519.4 |
|---|
| SMILES | CS(=O)(=O)Nc1ccc(CCNC(=O)c2ccc(O)c3nc([nH]c23)-c2ccc(Cl)cc2Cl)cc1 |
|---|
| Structure | 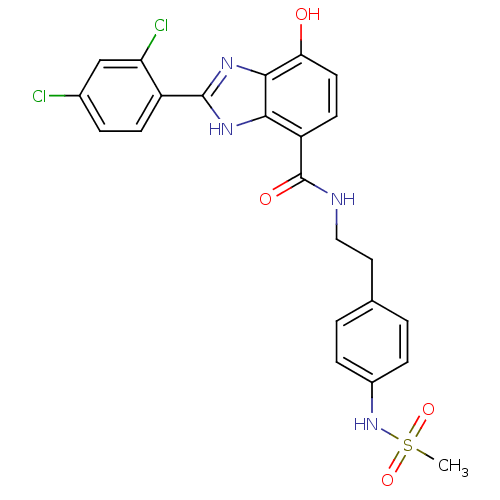
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shin, D; Lee, SC; Heo, YS; Lee, WY; Cho, YS; Kim, YE; Hyun, YL; Cho, JM; Lee, YS; Ro, S Design and synthesis of 7-hydroxy-1H-benzoimidazole derivatives as novel inhibitors of glycogen synthase kinase-3beta. Bioorg Med Chem Lett17:5686-9 (2007) [PubMed] Article
Shin, D; Lee, SC; Heo, YS; Lee, WY; Cho, YS; Kim, YE; Hyun, YL; Cho, JM; Lee, YS; Ro, S Design and synthesis of 7-hydroxy-1H-benzoimidazole derivatives as novel inhibitors of glycogen synthase kinase-3beta. Bioorg Med Chem Lett17:5686-9 (2007) [PubMed] Article