| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Disintegrin and metalloproteinase domain-containing protein 10 |
|---|
| Ligand | BDBM50229639 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_461861 (CHEMBL927854) |
|---|
| IC50 | 7.1±n/a nM |
|---|
| Citation |  Burns, DM; He, C; Li, Y; Scherle, P; Liu, X; Marando, CA; Covington, MB; Yang, G; Pan, M; Turner, S; Fridman, JS; Hollis, G; Vaddi, K; Yeleswaram, S; Newton, R; Friedman, S; Metcalf, B; Yao, W Conversion of an MMP-potent scaffold to an MMP-selective HER-2 sheddase inhibitor via scaffold hybridization and subtle P1' permutations. Bioorg Med Chem Lett18:560-4 (2008) [PubMed] Article Burns, DM; He, C; Li, Y; Scherle, P; Liu, X; Marando, CA; Covington, MB; Yang, G; Pan, M; Turner, S; Fridman, JS; Hollis, G; Vaddi, K; Yeleswaram, S; Newton, R; Friedman, S; Metcalf, B; Yao, W Conversion of an MMP-potent scaffold to an MMP-selective HER-2 sheddase inhibitor via scaffold hybridization and subtle P1' permutations. Bioorg Med Chem Lett18:560-4 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Disintegrin and metalloproteinase domain-containing protein 10 |
|---|
| Name: | Disintegrin and metalloproteinase domain-containing protein 10 |
|---|
| Synonyms: | ADA10_HUMAN | ADAM10 | Disintegrin and metalloproteinase domain-containing protein 10 (ADAM10) | KUZ | MADM |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 84160.93 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O14672 |
|---|
| Residue: | 748 |
|---|
| Sequence: | MVLLRVLILLLSWAAGMGGQYGNPLNKYIRHYEGLSYNVDSLHQKHQRAKRAVSHEDQFL
RLDFHAHGRHFNLRMKRDTSLFSDEFKVETSNKVLDYDTSHIYTGHIYGEEGSFSHGSVI
DGRFEGFIQTRGGTFYVEPAERYIKDRTLPFHSVIYHEDDINYPHKYGPQGGCADHSVFE
RMRKYQMTGVEEVTQIPQEEHAANGPELLRKKRTTSAEKNTCQLYIQTDHLFFKYYGTRE
AVIAQISSHVKAIDTIYQTTDFSGIRNISFMVKRIRINTTADEKDPTNPFRFPNIGVEKF
LELNSEQNHDDYCLAYVFTDRDFDDGVLGLAWVGAPSGSSGGICEKSKLYSDGKKKSLNT
GIITVQNYGSHVPPKVSHITFAHEVGHNFGSPHDSGTECTPGESKNLGQKENGNYIMYAR
ATSGDKLNNNKFSLCSIRNISQVLEKKRNNCFVESGQPICGNGMVEQGEECDCGYSDQCK
DECCFDANQPEGRKCKLKPGKQCSPSQGPCCTAQCAFKSKSEKCRDDSDCAREGICNGFT
ALCPASDPKPNFTDCNRHTQVCINGQCAGSICEKYGLEECTCASSDGKDDKELCHVCCMK
KMDPSTCASTGSVQWSRHFSGRTITLQPGSPCNDFRGYCDVFMRCRLVDADGPLARLKKA
IFSPELYENIAEWIVAHWWAVLLMGIALIMLMAGFIKICSVHTPSSNPKLPPPKPLPGTL
KRRRPPQPIQQPQRQRPRESYQMGHMRR
|
|
|
|---|
| BDBM50229639 |
|---|
| n/a |
|---|
| Name | BDBM50229639 |
|---|
| Synonyms: | (S)-tetrahydrofuran-3-yl 4-(hydroxycarbamoyl)-4-((4-phenyl-5,6-dihydropyridin-1(2H)-ylsulfonyl)methyl)piperidine-1-carboxylate | CHEMBL251820 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H31N3O7S |
|---|
| Mol. Mass. | 493.573 |
|---|
| SMILES | ONC(=O)C1(CS(=O)(=O)N2CCC(=CC2)c2ccccc2)CCN(CC1)C(=O)O[C@H]1CCOC1 |c:12| |
|---|
| Structure | 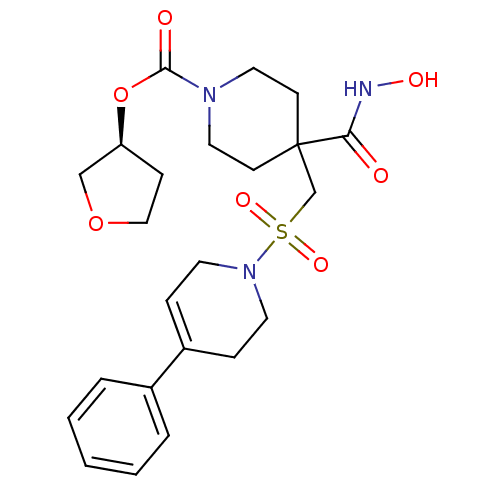
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Burns, DM; He, C; Li, Y; Scherle, P; Liu, X; Marando, CA; Covington, MB; Yang, G; Pan, M; Turner, S; Fridman, JS; Hollis, G; Vaddi, K; Yeleswaram, S; Newton, R; Friedman, S; Metcalf, B; Yao, W Conversion of an MMP-potent scaffold to an MMP-selective HER-2 sheddase inhibitor via scaffold hybridization and subtle P1' permutations. Bioorg Med Chem Lett18:560-4 (2008) [PubMed] Article
Burns, DM; He, C; Li, Y; Scherle, P; Liu, X; Marando, CA; Covington, MB; Yang, G; Pan, M; Turner, S; Fridman, JS; Hollis, G; Vaddi, K; Yeleswaram, S; Newton, R; Friedman, S; Metcalf, B; Yao, W Conversion of an MMP-potent scaffold to an MMP-selective HER-2 sheddase inhibitor via scaffold hybridization and subtle P1' permutations. Bioorg Med Chem Lett18:560-4 (2008) [PubMed] Article