| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50240380 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_497349 (CHEMBL999388) |
|---|
| IC50 | 900±n/a nM |
|---|
| Citation |  Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem43:1621-31 (2008) [PubMed] Article Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem43:1621-31 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50240380 |
|---|
| n/a |
|---|
| Name | BDBM50240380 |
|---|
| Synonyms: | 2,6-Bis-(benzylidene)-cyclohexanone | 2,6-Bis-[1-phenyl-meth-(E)-ylidene]-cyclohexanone | 2,6-dibenzylidenecyclohexanone | CHEMBL274993 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H18O |
|---|
| Mol. Mass. | 274.3563 |
|---|
| SMILES | O=C1\C(CCC\C1=C/c1ccccc1)=C\c1ccccc1 |
|---|
| Structure | 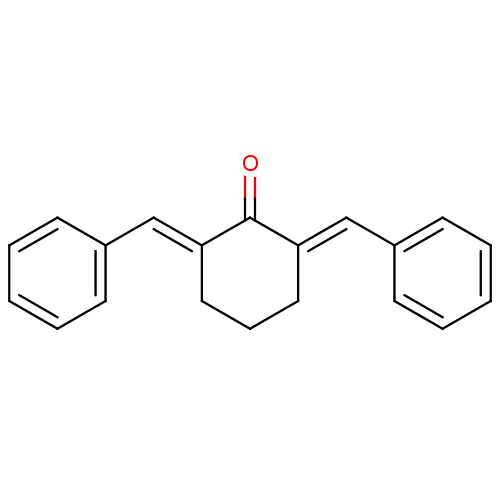
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem43:1621-31 (2008) [PubMed] Article
Appiah-Opong, R; de Esch, I; Commandeur, JN; Andarini, M; Vermeulen, NP Structure-activity relationships for the inhibition of recombinant human cytochromes P450 by curcumin analogues. Eur J Med Chem43:1621-31 (2008) [PubMed] Article