| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carboxylic ester hydrolase |
|---|
| Ligand | BDBM50261345 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_531228 (CHEMBL983416) |
|---|
| IC50 | 23±n/a nM |
|---|
| Citation |  Decker, M; Kraus, B; Heilmann, J Design, synthesis and pharmacological evaluation of hybrid molecules out of quinazolinimines and lipoic acid lead to highly potent and selective butyrylcholinesterase inhibitors with antioxidant properties. Bioorg Med Chem16:4252-61 (2008) [PubMed] Article Decker, M; Kraus, B; Heilmann, J Design, synthesis and pharmacological evaluation of hybrid molecules out of quinazolinimines and lipoic acid lead to highly potent and selective butyrylcholinesterase inhibitors with antioxidant properties. Bioorg Med Chem16:4252-61 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carboxylic ester hydrolase |
|---|
| Name: | Carboxylic ester hydrolase |
|---|
| Synonyms: | BuChE | Butyrlcholinesterase (BuChE) | Butyrylcholine esterase | Butyrylcholinesterase | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | Butyrylcholinesterase (EqBuChE) | Carboxylic ester hydrolase | butyrylcholinesterase precursor |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68842.83 |
|---|
| Organism: | Equus caballus (Horse) |
|---|
| Description: | Q9N1N9 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MQSWGTIICIRILLRFLLLWVLIGNSHTEEDIIITTKNGKVRGMNLPVLGGTVTAFLGIP
YAQPPLGRLRFKKPQSLTKWSNIWNATKYANSCYQNTDQSFPGFLGSEMWNPNTELSEDC
LYLNVWIPAPKPKNATVMIWIYGGGFQTGTSSLPVYDGKFLARVERVIVVSMNYRVGALG
FLALSENPEAPGNMGLFDQQLALQWVQKNIAAFGGNPRSVTLFGESAGAASVSLHLLSPR
SQPLFTRAILQSGSSNAPWAVTSLYEARNRTLTLAKRMGCSRDNETEMIKCLRDKDPQEI
LLNEVFVVPYDTLLSVNFGPTVDGDFLTDMPDTLLQLGQFKRTQILVGVNKDEGTAFLVY
GAPGFSKDNNSIITRKEFQEGLKIFFPRVSEFGRESILFHYMDWLDDQRAENYREALDDV
VGDYNIICPALEFTKKFSELGNDAFFYYFEHRSTKLPWPEWMGVMHGYEIEFVFGLPLER
RVNYTKAEEILSRSIMKRWANFAKYGNPNGTQSNSTRWPVFKSTEQKYLTLNTESPKVYT
KLRAQQCRFWTLFFPKVLELTGNIDEAEREWKAGFHRWNNYMMDWKNQFNDYTSKKESCS
DF
|
|
|
|---|
| BDBM50261345 |
|---|
| n/a |
|---|
| Name | BDBM50261345 |
|---|
| Synonyms: | 5-(1,2-Dithiolan-3-yl)-N-[8-(6,7,8,9,10,11-hexahydro-13H-azocino[2,1-b]quinazolin-13-ylideneamino)-octyl]pentanamide | CHEMBL466339 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H46N4OS2 |
|---|
| Mol. Mass. | 542.842 |
|---|
| SMILES | O=C(CCCCC1CCSS1)NCCCCCCCC\N=c1/n2CCCCCCc2nc2ccccc12 |
|---|
| Structure | 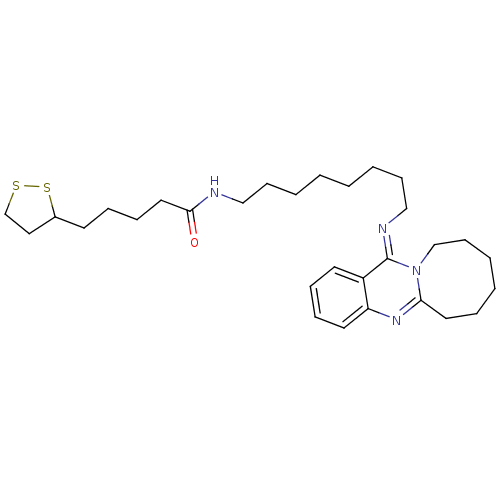
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Decker, M; Kraus, B; Heilmann, J Design, synthesis and pharmacological evaluation of hybrid molecules out of quinazolinimines and lipoic acid lead to highly potent and selective butyrylcholinesterase inhibitors with antioxidant properties. Bioorg Med Chem16:4252-61 (2008) [PubMed] Article
Decker, M; Kraus, B; Heilmann, J Design, synthesis and pharmacological evaluation of hybrid molecules out of quinazolinimines and lipoic acid lead to highly potent and selective butyrylcholinesterase inhibitors with antioxidant properties. Bioorg Med Chem16:4252-61 (2008) [PubMed] Article