| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tyrosine-protein kinase Lyn |
|---|
| Ligand | BDBM50274669 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_537189 (CHEMBL992565) |
|---|
| IC50 | >1000000±n/a nM |
|---|
| Citation |  Moriarty, KJ; Takahashi, H; Pullen, SS; Khine, HH; Sallati, RH; Raymond, EL; Woska, JR; Jeanfavre, DD; Roth, GP; Winters, MP; Qiao, L; Ryan, D; DesJarlais, R; Robinson, D; Wilson, M; Bobko, M; Cook, BN; Lo, HY; Nemoto, PA; Kashem, MA; Wolak, JP; White, A; Magolda, RL; Tomczuk, B Discovery, SAR and X-ray structure of 1H-benzimidazole-5-carboxylic acid cyclohexyl-methyl-amides as inhibitors of inducible T-cell kinase (Itk). Bioorg Med Chem Lett18:5545-9 (2008) [PubMed] Article Moriarty, KJ; Takahashi, H; Pullen, SS; Khine, HH; Sallati, RH; Raymond, EL; Woska, JR; Jeanfavre, DD; Roth, GP; Winters, MP; Qiao, L; Ryan, D; DesJarlais, R; Robinson, D; Wilson, M; Bobko, M; Cook, BN; Lo, HY; Nemoto, PA; Kashem, MA; Wolak, JP; White, A; Magolda, RL; Tomczuk, B Discovery, SAR and X-ray structure of 1H-benzimidazole-5-carboxylic acid cyclohexyl-methyl-amides as inhibitors of inducible T-cell kinase (Itk). Bioorg Med Chem Lett18:5545-9 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tyrosine-protein kinase Lyn |
|---|
| Name: | Tyrosine-protein kinase Lyn |
|---|
| Synonyms: | JTK8 | LYN | LYN_HUMAN | Lck/Yes-related novel protein tyrosine kinase | Tyrosine-protein kinase Lyn (LYN) | V-yes-1 Yamaguchi sarcoma viral related oncogene homolog | p53Lyn | p56Lyn |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58575.92 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P07948 |
|---|
| Residue: | 512 |
|---|
| Sequence: | MGCIKSKGKDSLSDDGVDLKTQPVRNTERTIYVRDPTSNKQQRPVPESQLLPGQRFQTKD
PEEQGDIVVALYPYDGIHPDDLSFKKGEKMKVLEEHGEWWKAKSLLTKKEGFIPSNYVAK
LNTLETEEWFFKDITRKDAERQLLAPGNSAGAFLIRESETLKGSFSLSVRDFDPVHGDVI
KHYKIRSLDNGGYYISPRITFPCISDMIKHYQKQADGLCRRLEKACISPKPQKPWDKDAW
EIPRESIKLVKRLGAGQFGEVWMGYYNNSTKVAVKTLKPGTMSVQAFLEEANLMKTLQHD
KLVRLYAVVTREEPIYIITEYMAKGSLLDFLKSDEGGKVLLPKLIDFSAQIAEGMAYIER
KNYIHRDLRAANVLVSESLMCKIADFGLARVIEDNEYTAREGAKFPIKWTAPEAINFGCF
TIKSDVWSFGILLYEIVTYGKIPYPGRTNADVMTALSQGYRMPRVENCPDELYDIMKMCW
KEKAEERPTFDYLQSVLDDFYTATEGQYQQQP
|
|
|
|---|
| BDBM50274669 |
|---|
| n/a |
|---|
| Name | BDBM50274669 |
|---|
| Synonyms: | CHEMBL485031 | N-(1-(3-(1H-imidazol-1-yl)propyl)-5-(N-methylcyclohexanecarboxamido)-1H-benzo[d]imidazol-2-yl)-4-cyanobenzamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H31N7O2 |
|---|
| Mol. Mass. | 509.6021 |
|---|
| SMILES | CN(C(=O)C1CCCCC1)c1ccc2n(CCCn3ccnc3)c(NC(=O)c3ccc(cc3)C#N)nc2c1 |
|---|
| Structure | 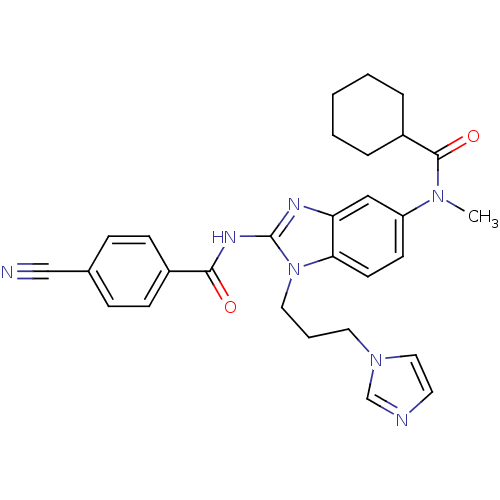
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Moriarty, KJ; Takahashi, H; Pullen, SS; Khine, HH; Sallati, RH; Raymond, EL; Woska, JR; Jeanfavre, DD; Roth, GP; Winters, MP; Qiao, L; Ryan, D; DesJarlais, R; Robinson, D; Wilson, M; Bobko, M; Cook, BN; Lo, HY; Nemoto, PA; Kashem, MA; Wolak, JP; White, A; Magolda, RL; Tomczuk, B Discovery, SAR and X-ray structure of 1H-benzimidazole-5-carboxylic acid cyclohexyl-methyl-amides as inhibitors of inducible T-cell kinase (Itk). Bioorg Med Chem Lett18:5545-9 (2008) [PubMed] Article
Moriarty, KJ; Takahashi, H; Pullen, SS; Khine, HH; Sallati, RH; Raymond, EL; Woska, JR; Jeanfavre, DD; Roth, GP; Winters, MP; Qiao, L; Ryan, D; DesJarlais, R; Robinson, D; Wilson, M; Bobko, M; Cook, BN; Lo, HY; Nemoto, PA; Kashem, MA; Wolak, JP; White, A; Magolda, RL; Tomczuk, B Discovery, SAR and X-ray structure of 1H-benzimidazole-5-carboxylic acid cyclohexyl-methyl-amides as inhibitors of inducible T-cell kinase (Itk). Bioorg Med Chem Lett18:5545-9 (2008) [PubMed] Article