

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cannabinoid receptor 2 | ||
| Ligand | BDBM50169954 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_564385 (CHEMBL962380) | ||
| Ki | 76±n/a nM | ||
| Citation |  Ferreira, AM; Krishnamurthy, M; Moore, BM; Finkelstein, D; Bashford, D Quantitative structure-activity relationship (QSAR) for a series of novel cannabinoid derivatives using descriptors derived from semi-empirical quantum-chemical calculations. Bioorg Med Chem17:2598-606 (2009) [PubMed] Article Ferreira, AM; Krishnamurthy, M; Moore, BM; Finkelstein, D; Bashford, D Quantitative structure-activity relationship (QSAR) for a series of novel cannabinoid derivatives using descriptors derived from semi-empirical quantum-chemical calculations. Bioorg Med Chem17:2598-606 (2009) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cannabinoid receptor 2 | |||
| Name: | Cannabinoid receptor 2 | ||
| Synonyms: | CANNABINOID CB2 | CB-2 | CB2 | CB2A | CB2B | CNR2 | CNR2_HUMAN | CX5 | Cannabinoid CB2 receptor | Cannabinoid receptor 2 (CB2) | Cannabinoid receptor 2 (CB2R) | hCB2 | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 39690.94 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P34972 | ||
| Residue: | 360 | ||
| Sequence: |
| ||
| BDBM50169954 | |||
| n/a | |||
| Name | BDBM50169954 | ||
| Synonyms: | (6aR,10aR)-3-Adamantan-2-ylmethyl-6,6,9-trimethyl-6a,7,10,10a-tetrahydro-6H-benzo[c]chromen-1-ol | AM-757 | CHEMBL372866 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C27H36O2 | ||
| Mol. Mass. | 392.5735 | ||
| SMILES | CC1=CC[C@@H]2[C@@H](C1)c1c(O)cc(CC3C4CC5CC(C4)CC3C5)cc1OC2(C)C |r,wU:5.7,wD:4.3,t:1,TLB:12:13:15:19.17.18,THB:17:16:13:19.18.20,17:18:15.16.22:13,20:18:15:22.21.13,20:21:15:19.17.18,(7.79,-4.59,;7.79,-6.13,;6.45,-6.92,;6.45,-8.46,;7.79,-9.23,;9.12,-8.46,;9.12,-6.92,;10.46,-9.23,;11.78,-8.47,;11.79,-6.92,;13.11,-9.24,;13.1,-10.78,;14.44,-11.56,;15.78,-10.8,;15.79,-9.31,;14.59,-8.03,;16.1,-8.45,;17.51,-7.88,;18.53,-9.16,;17.12,-8.81,;18.54,-10.7,;17.14,-11.28,;16.08,-10.04,;11.76,-11.54,;10.44,-10.76,;9.11,-11.54,;7.79,-10.76,;6.28,-11.16,;7.38,-12.25,)| | ||
| Structure | 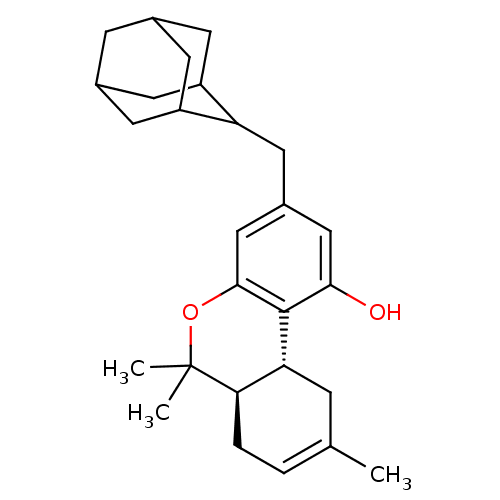
| ||