| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor gamma |
|---|
| Ligand | BDBM50278205 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_565041 (CHEMBL959184) |
|---|
| EC50 | 4000±n/a nM |
|---|
| Citation |  Otero, MP; Pérez Santín, E; Rodríguez-Barrios, F; Vaz, B; de Lera, AR Selective, potent PPARgamma agonists with cyclopentenone core structure. Bioorg Med Chem Lett19:1883-6 (2009) [PubMed] Article Otero, MP; Pérez Santín, E; Rodríguez-Barrios, F; Vaz, B; de Lera, AR Selective, potent PPARgamma agonists with cyclopentenone core structure. Bioorg Med Chem Lett19:1883-6 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor gamma |
|---|
| Name: | Peroxisome proliferator-activated receptor gamma |
|---|
| Synonyms: | NR1C3 | Nuclear receptor subfamily 1 group C member 3 | PPAR-gamma | PPARG | PPARG_HUMAN | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor gamma (PPAR gamma) | Peroxisome proliferator-activated receptor gamma (PPARG) | Peroxisome proliferator-activated receptor gamma (PPARγ) | Peroxisome proliferator-activated receptor gamma/Nuclear receptor corepressor 2 | peroxisome proliferator-activated receptor gamma isoform 2 |
|---|
| Type: | Nuclear Receptor |
|---|
| Mol. Mass.: | 57613.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P37231 |
|---|
| Residue: | 505 |
|---|
| Sequence: | MGETLGDSPIDPESDSFTDTLSANISQEMTMVDTEMPFWPTNFGISSVDLSVMEDHSHSF
DIKPFTTVDFSSISTPHYEDIPFTRTDPVVADYKYDLKLQEYQSAIKVEPASPPYYSEKT
QLYNKPHEEPSNSLMAIECRVCGDKASGFHYGVHACEGCKGFFRRTIRLKLIYDRCDLNC
RIHKKSRNKCQYCRFQKCLAVGMSHNAIRFGRMPQAEKEKLLAEISSDIDQLNPESADLR
ALAKHLYDSYIKSFPLTKAKARAILTGKTTDKSPFVIYDMNSLMMGEDKIKFKHITPLQE
QSKEVAIRIFQGCQFRSVEAVQEITEYAKSIPGFVNLDLNDQVTLLKYGVHEIIYTMLAS
LMNKDGVLISEGQGFMTREFLKSLRKPFGDFMEPKFEFAVKFNALELDDSDLAIFIAVII
LSGDRPGLLNVKPIEDIQDNLLQALELQLKLNHPESSQLFAKLLQKMTDLRQIVTEHVQL
LQVIKKTETDMSLHPLLQEIYKDLY
|
|
|
|---|
| BDBM50278205 |
|---|
| n/a |
|---|
| Name | BDBM50278205 |
|---|
| Synonyms: | 6-((1S,5R)-5-heptyl-4-oxocyclopent-2-enyloxy)hexanoic acid | CHEMBL472071 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H30O4 |
|---|
| Mol. Mass. | 310.4284 |
|---|
| SMILES | CCCCCCC[C@@H]1[C@@H](OCCCCCC(O)=O)C=CC1=O |r,c:18| |
|---|
| Structure | 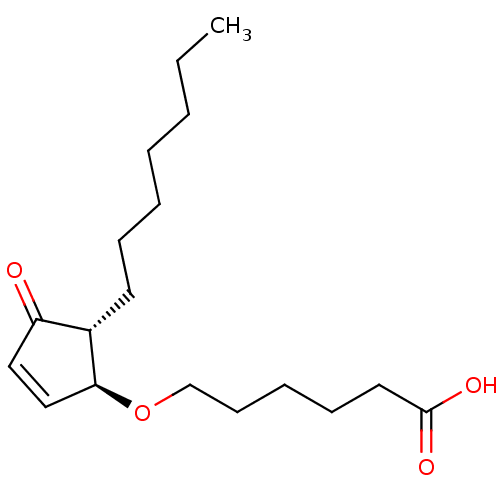
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Otero, MP; Pérez Santín, E; Rodríguez-Barrios, F; Vaz, B; de Lera, AR Selective, potent PPARgamma agonists with cyclopentenone core structure. Bioorg Med Chem Lett19:1883-6 (2009) [PubMed] Article
Otero, MP; Pérez Santín, E; Rodríguez-Barrios, F; Vaz, B; de Lera, AR Selective, potent PPARgamma agonists with cyclopentenone core structure. Bioorg Med Chem Lett19:1883-6 (2009) [PubMed] Article