| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Carbonic anhydrase |
|---|
| Ligand | BDBM50258678 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_519732 (CHEMBL938995) |
|---|
| Ki | 77±n/a nM |
|---|
| Citation |  Güzel, O; Maresca, A; Scozzafava, A; Salman, A; Balaban, AT; Supuran, CT Discovery of low nanomolar and subnanomolar inhibitors of the mycobacterial beta-carbonic anhydrases Rv1284 and Rv3273. J Med Chem52:4063-7 (2009) [PubMed] Article Güzel, O; Maresca, A; Scozzafava, A; Salman, A; Balaban, AT; Supuran, CT Discovery of low nanomolar and subnanomolar inhibitors of the mycobacterial beta-carbonic anhydrases Rv1284 and Rv3273. J Med Chem52:4063-7 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Carbonic anhydrase |
|---|
| Name: | Carbonic anhydrase |
|---|
| Synonyms: | PROBABLE TRANSMEMBRANE CARBONIC ANHYDRASE (CARBONATE DEHYDRATASE) (CARBONIC DEHYDRATASE) |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 80567.22 |
|---|
| Organism: | Mycobacterium tuberculosis |
|---|
| Description: | ChEMBL_878775 |
|---|
| Residue: | 764 |
|---|
| Sequence: | MTIPRSQHMSTAVNSCTEAPASRSQWMLANLRHDVPASLVVFLVALPLSLGIAIASGAPI
IAGVIAAVVGGIVAGAVGGSPVQVSGPAAGLTVVVAELIDELGWPMLCLMTIAAGALQIV
FGLSRMARAALAIAPVVVHAMLAGIGITIALQQIHVLLGGTSHSSAWRNIVALPDGILHH
ELHEVIVGGTVIAILLMWSKLPAKVRIIPGPLVAIAGATVLALLPVLQTERIDLQGNFFD
AIGLPKLAEMSPGGQPWSHEISAIALGVLTIALIASVESLLSAVGVDKLHHGPRTDFNRE
MVGQGSANVVSGLLGGLPITGVIVRSSANVAAGARTRMSTILHGVWILLFASLFTNLVEL
IPKAALAGLLIVIGAQLVKLAHIKLAWRTGNFVIYAITIVCVVFLNLLEGVAIGLVVAIV
FLLVRVVRAPVEVKPVGGEQSKRWRVDIDGTLSFLLLPRLTTVLSKLPEGSEVTLNLNAD
YIDDSVSEAISDWRRAHETRGGVVAIVETSPAKLHHAHARPPKRHFASDPIGLVPWRSAR
GKDRGSASVLDRIDEYHRNGAAVLHPHIAGLTDSQDPYELFLTCADSRILPNVITASGPG
DLYTVRNLGNLVPTDPDDRSVDAALDFAVNQLGVSSVVVCGHSSCAAMTALLEDDPANTT
TPMMRWLENAHDSLVVFRNHHPARRSAESAGYPEADQLSIVNVAVQVERLTRHPILATAV
AAADLQVIGIFFDISTARVYEVGPNGIICPDEPADRPVDHESAQ
|
|
|
|---|
| BDBM50258678 |
|---|
| n/a |
|---|
| Name | BDBM50258678 |
|---|
| Synonyms: | 1-(3-(4-chlorophenyl)-5-sulfamoyl-1H-indole-2-carboxamido)-2,4,6-trimethylpyridinium perchlorate | 1-({[5-(Aminosulfonyl)-3-(4-fluorophenyl)-1H-indol-2-yl]carbonyl}amino)-2,4,6 trimethylpyridinium perchlorate | CHEMBL513854 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H22ClN4O3S |
|---|
| Mol. Mass. | 469.963 |
|---|
| SMILES | Cc1cc(C)[n+](NC(=O)c2[nH]c3ccc(cc3c2-c2ccc(Cl)cc2)S(N)(=O)=O)c(C)c1 |
|---|
| Structure | 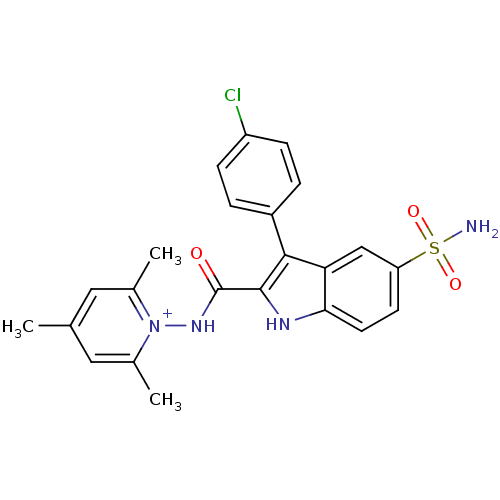
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Güzel, O; Maresca, A; Scozzafava, A; Salman, A; Balaban, AT; Supuran, CT Discovery of low nanomolar and subnanomolar inhibitors of the mycobacterial beta-carbonic anhydrases Rv1284 and Rv3273. J Med Chem52:4063-7 (2009) [PubMed] Article
Güzel, O; Maresca, A; Scozzafava, A; Salman, A; Balaban, AT; Supuran, CT Discovery of low nanomolar and subnanomolar inhibitors of the mycobacterial beta-carbonic anhydrases Rv1284 and Rv3273. J Med Chem52:4063-7 (2009) [PubMed] Article