| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Ligand | BDBM50279277 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_546016 (CHEMBL1028494) |
|---|
| Ki | 2250±n/a nM |
|---|
| Citation |  Tasso, B; Canu Boido, C; Terranova, E; Gotti, C; Riganti, L; Clementi, F; Artali, R; Bombieri, G; Meneghetti, F; Sparatore, F Synthesis, binding, and modeling studies of new cytisine derivatives, as ligands for neuronal nicotinic acetylcholine receptor subtypes. J Med Chem52:4345-57 (2009) [PubMed] Article Tasso, B; Canu Boido, C; Terranova, E; Gotti, C; Riganti, L; Clementi, F; Artali, R; Bombieri, G; Meneghetti, F; Sparatore, F Synthesis, binding, and modeling studies of new cytisine derivatives, as ligands for neuronal nicotinic acetylcholine receptor subtypes. J Med Chem52:4345-57 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Name: | Neuronal acetylcholine receptor subunit alpha-7 |
|---|
| Synonyms: | ACHA7_RAT | Acra7 | Cholinergic, Nicotinic Alpha7 | Cholinergic, Nicotinic Alpha7/5-HT3 | Chrna7 | Neuronal acetylcholine receptor | Neuronal acetylcholine receptor (alpha7 nAChR) | Neuronal acetylcholine receptor subunit alpha 7 | Neuronal acetylcholine receptor subunit alpha-7 | Neuronal acetylcholine receptor subunit alpha-7 (nAChR alpha7) | Neuronal acetylcholine receptor subunit alpha-7 (nAChR) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56502.44 |
|---|
| Organism: | Rattus norvegicus (Rat) |
|---|
| Description: | Q05941 |
|---|
| Residue: | 502 |
|---|
| Sequence: | MCGGRGGIWLALAAALLHVSLQGEFQRRLYKELVKNYNPLERPVANDSQPLTVYFSLSLL
QIMDVDEKNQVLTTNIWLQMSWTDHYLQWNMSEYPGVKNVRFPDGQIWKPDILLYNSADE
RFDATFHTNVLVNASGHCQYLPPGIFKSSCYIDVRWFPFDVQQCKLKFGSWSYGGWSLDL
QMQEADISSYIPNGEWDLMGIPGKRNEKFYECCKEPYPDVTYTVTMRRRTLYYGLNLLIP
CVLISALALLVFLLPADSGEKISLGITVLLSLTVFMLLVAEIMPATSDSVPLIAQYFAST
MIIVGLSVVVTVIVLRYHHHDPDGGKMPKWTRIILLNWCAWFLRMKRPGEDKVRPACQHK
PRRCSLASVELSAGAGPPTSNGNLLYIGFRGLEGMHCAPTPDSGVVCGRLACSPTHDEHL
MHGAHPSDGDPDLAKILEEVRYIANRFRCQDESEVICSEWKFAACVVDRLCLMAFSVFTI
ICTIGILMSAPNFVEAVSKDFA
|
|
|
|---|
| BDBM50279277 |
|---|
| n/a |
|---|
| Name | BDBM50279277 |
|---|
| Synonyms: | 1-Phenyl-2-(cytisin-12-yl)-1-ethanone | CHEMBL502029 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H20N2O2 |
|---|
| Mol. Mass. | 308.3743 |
|---|
| SMILES | O=C(CN1C[C@@H]2C[C@H](C1)c1cccc(=O)n1C2)c1ccccc1 |r| |
|---|
| Structure | 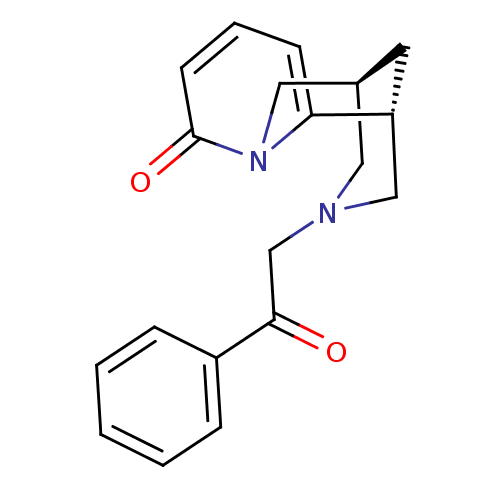
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tasso, B; Canu Boido, C; Terranova, E; Gotti, C; Riganti, L; Clementi, F; Artali, R; Bombieri, G; Meneghetti, F; Sparatore, F Synthesis, binding, and modeling studies of new cytisine derivatives, as ligands for neuronal nicotinic acetylcholine receptor subtypes. J Med Chem52:4345-57 (2009) [PubMed] Article
Tasso, B; Canu Boido, C; Terranova, E; Gotti, C; Riganti, L; Clementi, F; Artali, R; Bombieri, G; Meneghetti, F; Sparatore, F Synthesis, binding, and modeling studies of new cytisine derivatives, as ligands for neuronal nicotinic acetylcholine receptor subtypes. J Med Chem52:4345-57 (2009) [PubMed] Article