| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dopamine beta-hydroxylase |
|---|
| Ligand | BDBM50286815 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_59304 |
|---|
| Ki | 150000±n/a nM |
|---|
| Citation |  Eydoux, F; Chlenov, MA; Réglier, M Synthesis of indane derivatives as mechanism-based inhibitors of dopamine β-hydroxylase Bioorg Med Chem Lett5:941-944 (1995) Article Eydoux, F; Chlenov, MA; Réglier, M Synthesis of indane derivatives as mechanism-based inhibitors of dopamine β-hydroxylase Bioorg Med Chem Lett5:941-944 (1995) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Dopamine beta-hydroxylase |
|---|
| Name: | Dopamine beta-hydroxylase |
|---|
| Synonyms: | DBH | DOPO_BOVIN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 68136.39 |
|---|
| Organism: | Bos taurus |
|---|
| Description: | ChEMBL_50738 |
|---|
| Residue: | 610 |
|---|
| Sequence: | MQVPSPSVREAASMYGTAVAVFLVILVAALQGSAPAESPFPFHIPLDPEGTLELSWNISY
AQETIYFQLLVRELKAGVLFGMSDRGELENADLVVLWTDRDGAYFGDAWSDQKGQVHLDS
QQDYQLLRAQRTPEGLYLLFKRPFGTCDPNDYLIEDGTVHLVYGFLEEPLRSLESINTSG
LHTGLQRVQLLKPSIPKPALPADTRTMEIRAPDVLIPGQQTTYWCYVTELPDGFPRHHIV
MYEPIVTEGNEALVHHMEVFQCAAEFETIPHFSGPCDSKMKPQRLNFCRHVLAAWALGAK
AFYYPEEAGLAFGGPGSSRFLRLEVHYHNPLVITGRRDSSGIRLYYTAALRRFDAGIMEL
GLAYTPVMAIPPQETAFVLTGYCTDKCTQLALPASGIHIFASQLHTHLTGRKVVTVLARD
GRETEIVNRDNHYSPHFQEIRMLKKVVSVQPGDVLITSCTYNTEDRRLATVGGFGILEEM
CVNYVHYYPQTQLELCKSAVDPGFLHKYFRLVNRFNSEEVCTCPQASVPEQFASVPWNSF
NREVLKALYGFAPISMHCNRSSAVRFQGEWNRQPLPEIVSRLEEPTPHCPASQAQSPAGP
TVLNISGGKG
|
|
|
|---|
| BDBM50286815 |
|---|
| n/a |
|---|
| Name | BDBM50286815 |
|---|
| Synonyms: | 1,1a,6,6a-Tetrahydro-cyclopropa[a]inden-3-ol | CHEMBL368520 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C10H10O |
|---|
| Mol. Mass. | 146.1858 |
|---|
| SMILES | Oc1ccc2CC3CC3c2c1 |
|---|
| Structure | 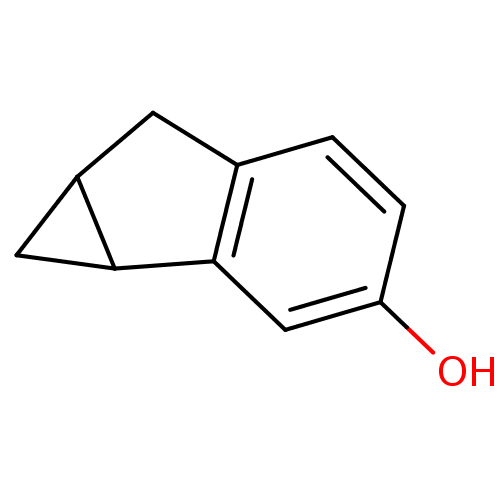
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Eydoux, F; Chlenov, MA; Réglier, M Synthesis of indane derivatives as mechanism-based inhibitors of dopamine β-hydroxylase Bioorg Med Chem Lett5:941-944 (1995) Article
Eydoux, F; Chlenov, MA; Réglier, M Synthesis of indane derivatives as mechanism-based inhibitors of dopamine β-hydroxylase Bioorg Med Chem Lett5:941-944 (1995) Article