| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cholecystokinin receptor type A |
|---|
| Ligand | BDBM50286896 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_50038 |
|---|
| IC50 | 433±n/a nM |
|---|
| Citation |  Semple, G; Ryder, H; Kendrick, DA; Szelke, M; Ohta, M; Satoh, M; Nishida, A; Akuzawa, S; Miyata, K Synthesis and biological activity of 5-heteroaryl benzodiazepines: analogues of YM022 Bioorg Med Chem Lett6:55-58 (1996) Article Semple, G; Ryder, H; Kendrick, DA; Szelke, M; Ohta, M; Satoh, M; Nishida, A; Akuzawa, S; Miyata, K Synthesis and biological activity of 5-heteroaryl benzodiazepines: analogues of YM022 Bioorg Med Chem Lett6:55-58 (1996) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cholecystokinin receptor type A |
|---|
| Name: | Cholecystokinin receptor type A |
|---|
| Synonyms: | CCKAR_RAT | Cckar | Cholecystokinin peripheral | Cholecystokinin receptor | Cholecystokinin receptor type A |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 49676.37 |
|---|
| Organism: | RAT |
|---|
| Description: | Cholecystokinin central 0 RAT::P30551 |
|---|
| Residue: | 444 |
|---|
| Sequence: | MSHSPARQHLVESSRMDVVDSLLMNGSNITPPCELGLENETLFCLDQPQPSKEWQSALQI
LLYSIIFLLSVLGNTLVITVLIRNKRMRTVTNIFLLSLAVSDLMLCLFCMPFNLIPNLLK
DFIFGSAVCKTTTYFMGTSVSVSTFNLVAISLERYGAICRPLQSRVWQTKSHALKVIAAT
WCLSFTIMTPYPIYSNLVPFTKNNNQTANMCRFLLPSDAMQQSWQTFLLLILFLLPGIVM
VVAYGLISLELYQGIKFDASQKKSAKEKKPSTGSSTRYEDSDGCYLQKSRPPRKLELQQL
SSGSGGSRLNRIRSSSSAANLIAKKRVIRMLIVIVVLFFLCWMPIFSANAWRAYDTVSAE
KHLSGTPISFILLLSYTSSCVNPIIYCFMNKRFRLGFMATFPCCPNPGPPGVRGEVGEEE
DGRTIRALLSRYSYSHMSTSAPPP
|
|
|
|---|
| BDBM50286896 |
|---|
| n/a |
|---|
| Name | BDBM50286896 |
|---|
| Synonyms: | 1-(3,3-Dimethyl-2-oxo-butyl)-5-thiazol-2-yl-3-[(m-tolylamino-methyl)-amino]-1,3-dihydro-benzo[e][1,4]diazepin-2-one | CHEMBL435447 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29N5O2S |
|---|
| Mol. Mass. | 475.606 |
|---|
| SMILES | Cc1cccc(NCNC2N=C(c3nccs3)c3ccccc3N(CC(=O)C(C)(C)C)C2=O)c1 |t:10| |
|---|
| Structure | 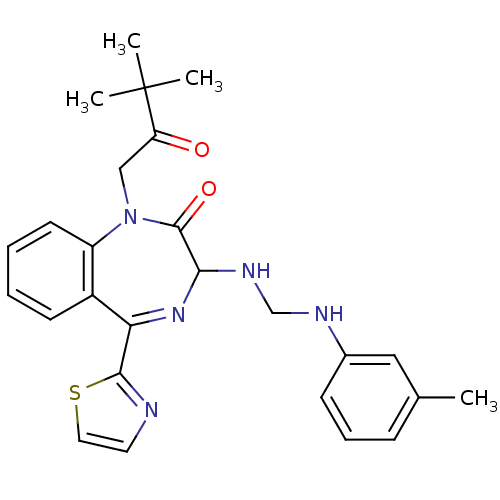
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Semple, G; Ryder, H; Kendrick, DA; Szelke, M; Ohta, M; Satoh, M; Nishida, A; Akuzawa, S; Miyata, K Synthesis and biological activity of 5-heteroaryl benzodiazepines: analogues of YM022 Bioorg Med Chem Lett6:55-58 (1996) Article
Semple, G; Ryder, H; Kendrick, DA; Szelke, M; Ohta, M; Satoh, M; Nishida, A; Akuzawa, S; Miyata, K Synthesis and biological activity of 5-heteroaryl benzodiazepines: analogues of YM022 Bioorg Med Chem Lett6:55-58 (1996) Article