| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prothrombin |
|---|
| Ligand | BDBM50287476 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_208346 (CHEMBL813656) |
|---|
| Ki | 0.190000±n/a nM |
|---|
| Citation |  Steinmetzer, T; Konishi, Y Tripeptidyl pyridinium methyl ketones as potent active site inhibitors of thrombin Bioorg Med Chem Lett6:1677-1682 (1996) Article Steinmetzer, T; Konishi, Y Tripeptidyl pyridinium methyl ketones as potent active site inhibitors of thrombin Bioorg Med Chem Lett6:1677-1682 (1996) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prothrombin |
|---|
| Name: | Prothrombin |
|---|
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 70029.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P00734 |
|---|
| Residue: | 622 |
|---|
| Sequence: | MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLEREC
VEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHV
NITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQE
CSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASA
QAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETG
DGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYI
DGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTEN
DLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHP
VCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDST
RIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKY
GFYTHVFRLKKWIQKVIDQFGE
|
|
|
|---|
| BDBM50287476 |
|---|
| n/a |
|---|
| Name | BDBM50287476 |
|---|
| Synonyms: | 1-((S)-3-{[(S)-1-((R)-2-Amino-3-cyclohexyl-propionyl)-pyrrolidine-2-carbonyl]-amino}-6-guanidino-2-oxo-hexyl)-pyridinium | CHEMBL295863 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H42N7O3 |
|---|
| Mol. Mass. | 500.6562 |
|---|
| SMILES | N[C@H](CC1CCCCC1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)C[n+]1ccccc1 |
|---|
| Structure | 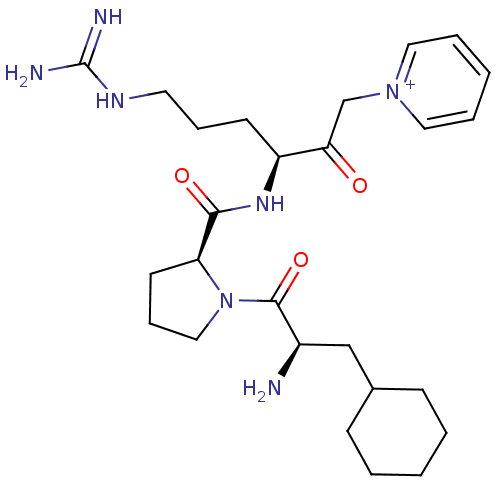
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Steinmetzer, T; Konishi, Y Tripeptidyl pyridinium methyl ketones as potent active site inhibitors of thrombin Bioorg Med Chem Lett6:1677-1682 (1996) Article
Steinmetzer, T; Konishi, Y Tripeptidyl pyridinium methyl ketones as potent active site inhibitors of thrombin Bioorg Med Chem Lett6:1677-1682 (1996) Article